Input interpretation
octanoic acid, lead(2+) salt | samarium(III) fluoride
Chemical names and formulas
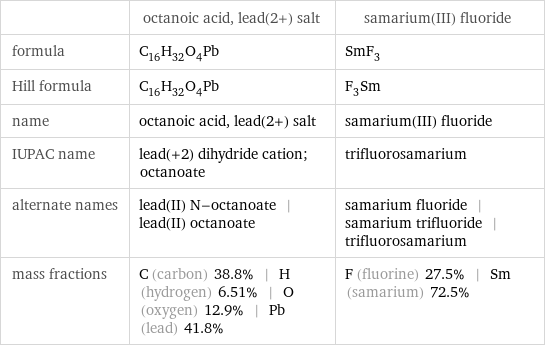
| octanoic acid, lead(2+) salt | samarium(III) fluoride formula | C_16H_32O_4Pb | SmF_3 Hill formula | C_16H_32O_4Pb | F_3Sm name | octanoic acid, lead(2+) salt | samarium(III) fluoride IUPAC name | lead(+2) dihydride cation; octanoate | trifluorosamarium alternate names | lead(II) N-octanoate | lead(II) octanoate | samarium fluoride | samarium trifluoride | trifluorosamarium mass fractions | C (carbon) 38.8% | H (hydrogen) 6.51% | O (oxygen) 12.9% | Pb (lead) 41.8% | F (fluorine) 27.5% | Sm (samarium) 72.5%
Structure diagrams
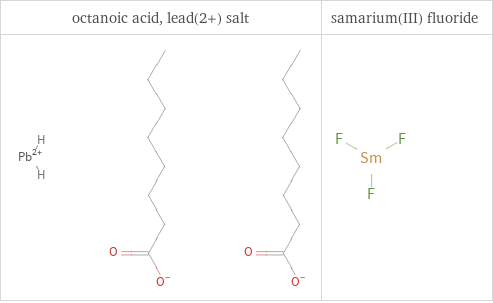
Structure diagrams
Basic properties
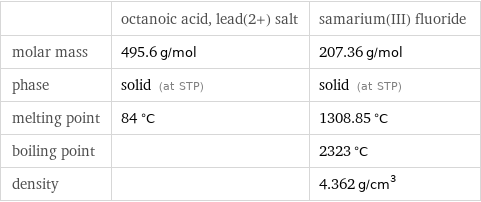
| octanoic acid, lead(2+) salt | samarium(III) fluoride molar mass | 495.6 g/mol | 207.36 g/mol phase | solid (at STP) | solid (at STP) melting point | 84 °C | 1308.85 °C boiling point | | 2323 °C density | | 4.362 g/cm^3
Units
