Input interpretation
mercury(II) cation | zinc cation | barium cation
Structure diagrams
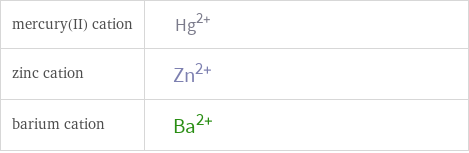
Structure diagrams
General properties
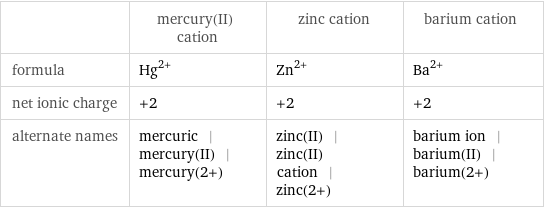
| mercury(II) cation | zinc cation | barium cation formula | Hg^(2+) | Zn^(2+) | Ba^(2+) net ionic charge | +2 | +2 | +2 alternate names | mercuric | mercury(II) | mercury(2+) | zinc(II) | zinc(II) cation | zinc(2+) | barium ion | barium(II) | barium(2+)
Ionic radii
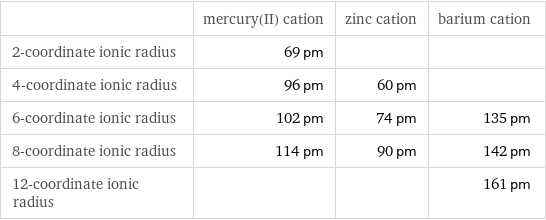
| mercury(II) cation | zinc cation | barium cation 2-coordinate ionic radius | 69 pm | | 4-coordinate ionic radius | 96 pm | 60 pm | 6-coordinate ionic radius | 102 pm | 74 pm | 135 pm 8-coordinate ionic radius | 114 pm | 90 pm | 142 pm 12-coordinate ionic radius | | | 161 pm
Units

Other properties
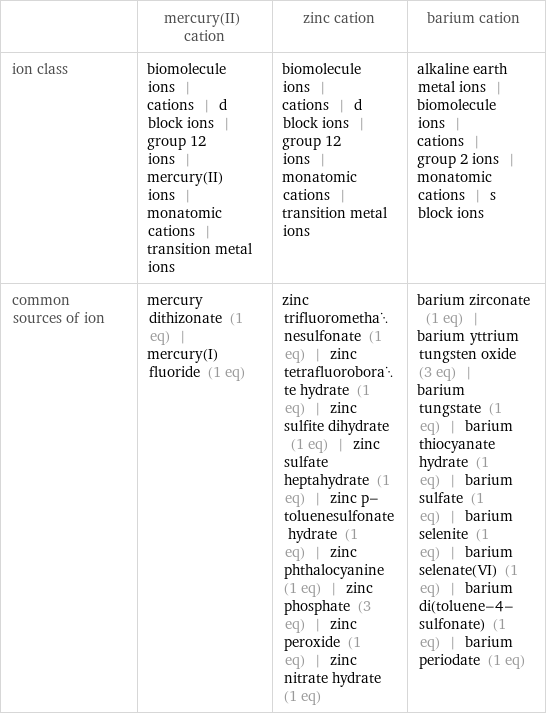
| mercury(II) cation | zinc cation | barium cation ion class | biomolecule ions | cations | d block ions | group 12 ions | mercury(II) ions | monatomic cations | transition metal ions | biomolecule ions | cations | d block ions | group 12 ions | monatomic cations | transition metal ions | alkaline earth metal ions | biomolecule ions | cations | group 2 ions | monatomic cations | s block ions common sources of ion | mercury dithizonate (1 eq) | mercury(I) fluoride (1 eq) | zinc trifluoromethanesulfonate (1 eq) | zinc tetrafluoroborate hydrate (1 eq) | zinc sulfite dihydrate (1 eq) | zinc sulfate heptahydrate (1 eq) | zinc p-toluenesulfonate hydrate (1 eq) | zinc phthalocyanine (1 eq) | zinc phosphate (3 eq) | zinc peroxide (1 eq) | zinc nitrate hydrate (1 eq) | barium zirconate (1 eq) | barium yttrium tungsten oxide (3 eq) | barium tungstate (1 eq) | barium thiocyanate hydrate (1 eq) | barium sulfate (1 eq) | barium selenite (1 eq) | barium selenate(VI) (1 eq) | barium di(toluene-4-sulfonate) (1 eq) | barium periodate (1 eq)