Input interpretation
hexabromoplatinate(IV) anion | carbonate anion | diphosphate anion
Structure diagrams
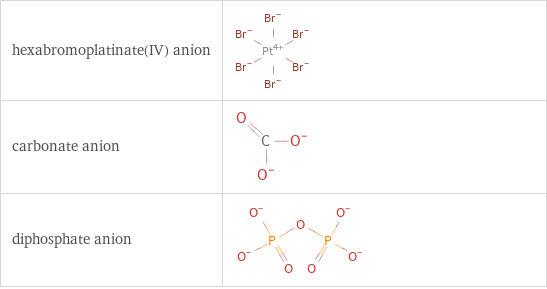
Structure diagrams
General properties
![| hexabromoplatinate(IV) anion | carbonate anion | diphosphate anion formula | ([PtBr_6])^(2-) | (CO_3)^(2-) | (P_2O_7)^(4-) net ionic charge | -2 | -2 | -4 alternate names | hexabromoplatinate(IV) | hexabromoplatinate(2-) | trioxocarbonate | trioxidocarbonate | carbonate | carbonate(2-) | pyrophosphate | diphosphate | diphosphate(4-)](../image_source/75bee6272d5c1fac5443491460c91e4c.png)
| hexabromoplatinate(IV) anion | carbonate anion | diphosphate anion formula | ([PtBr_6])^(2-) | (CO_3)^(2-) | (P_2O_7)^(4-) net ionic charge | -2 | -2 | -4 alternate names | hexabromoplatinate(IV) | hexabromoplatinate(2-) | trioxocarbonate | trioxidocarbonate | carbonate | carbonate(2-) | pyrophosphate | diphosphate | diphosphate(4-)
Ionic radii

| hexabromoplatinate(IV) anion | carbonate anion thermochemical radius | 342 pm | 178 pm
Units

Other properties
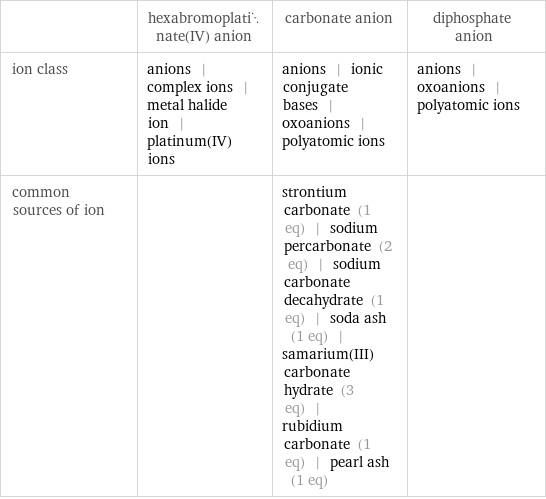
| hexabromoplatinate(IV) anion | carbonate anion | diphosphate anion ion class | anions | complex ions | metal halide ion | platinum(IV) ions | anions | ionic conjugate bases | oxoanions | polyatomic ions | anions | oxoanions | polyatomic ions common sources of ion | | strontium carbonate (1 eq) | sodium percarbonate (2 eq) | sodium carbonate decahydrate (1 eq) | soda ash (1 eq) | samarium(III) carbonate hydrate (3 eq) | rubidium carbonate (1 eq) | pearl ash (1 eq) |