Input interpretation
ammonium chloride | silver perchlorate hydrate
Chemical names and formulas
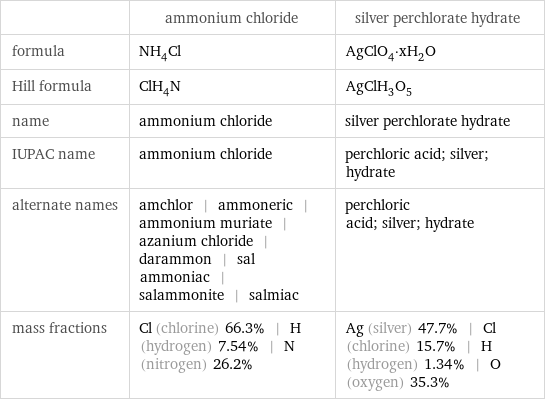
| ammonium chloride | silver perchlorate hydrate formula | NH_4Cl | AgClO_4·xH_2O Hill formula | ClH_4N | AgClH_3O_5 name | ammonium chloride | silver perchlorate hydrate IUPAC name | ammonium chloride | perchloric acid; silver; hydrate alternate names | amchlor | ammoneric | ammonium muriate | azanium chloride | darammon | sal ammoniac | salammonite | salmiac | perchloric acid; silver; hydrate mass fractions | Cl (chlorine) 66.3% | H (hydrogen) 7.54% | N (nitrogen) 26.2% | Ag (silver) 47.7% | Cl (chlorine) 15.7% | H (hydrogen) 1.34% | O (oxygen) 35.3%
Structure diagrams
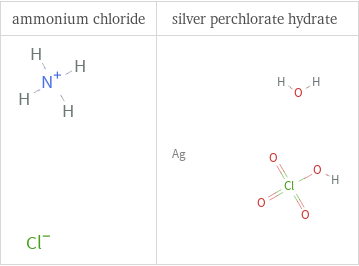
Structure diagrams
Basic properties
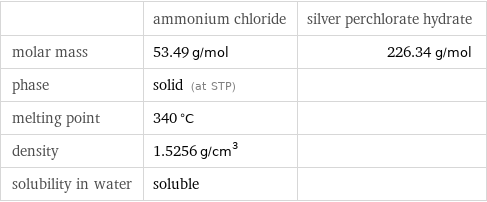
| ammonium chloride | silver perchlorate hydrate molar mass | 53.49 g/mol | 226.34 g/mol phase | solid (at STP) | melting point | 340 °C | density | 1.5256 g/cm^3 | solubility in water | soluble |
Units
