Input interpretation
hydrogen fluoride | hydrogen bromide
Chemical names and formulas
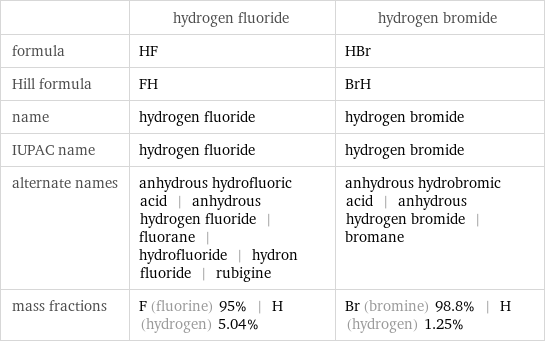
| hydrogen fluoride | hydrogen bromide formula | HF | HBr Hill formula | FH | BrH name | hydrogen fluoride | hydrogen bromide IUPAC name | hydrogen fluoride | hydrogen bromide alternate names | anhydrous hydrofluoric acid | anhydrous hydrogen fluoride | fluorane | hydrofluoride | hydron fluoride | rubigine | anhydrous hydrobromic acid | anhydrous hydrogen bromide | bromane mass fractions | F (fluorine) 95% | H (hydrogen) 5.04% | Br (bromine) 98.8% | H (hydrogen) 1.25%
Structure diagrams

Structure diagrams
3D structure
3D structure
Basic properties
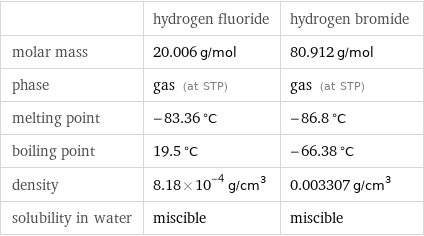
| hydrogen fluoride | hydrogen bromide molar mass | 20.006 g/mol | 80.912 g/mol phase | gas (at STP) | gas (at STP) melting point | -83.36 °C | -86.8 °C boiling point | 19.5 °C | -66.38 °C density | 8.18×10^-4 g/cm^3 | 0.003307 g/cm^3 solubility in water | miscible | miscible
Units

Gas properties (at STP)
| hydrogen fluoride | hydrogen bromide density | 8.18×10^-4 g/cm^3 | 0.003307 g/cm^3 vapor density | 1.27 | 2.71 molar volume | 24500 cm^3/mol | 24470 cm^3/mol surface tension | | 0.0271 N/m refractive index | 1.00001 | 1.325 dynamic viscosity | 1.2571×10^-5 Pa s | 8.4×10^-4 Pa s
Units

Liquid properties

| hydrogen bromide sound speed | 720 km/h
Units

Thermodynamic properties
| hydrogen fluoride | hydrogen bromide molar heat of vaporization | 7.5 kJ/mol (kilojoules per mole) | 17.3 kJ/mol (kilojoules per mole) specific heat of vaporization | 0.37 kJ/g (kilojoules per gram) | 0.214 kJ/g (kilojoules per gram) molar heat of fusion | 4.58 kJ/mol (kilojoules per mole) | 2.41 kJ/mol (kilojoules per mole) specific heat of fusion | 0.229 kJ/g (kilojoules per gram) | 0.0298 kJ/g (kilojoules per gram) critical temperature | 461 K (kelvins) | 363 K (kelvins) critical pressure | 6.48 MPa (megapascals) | 8.5 MPa (megapascals) acentric factor ω | 0.383 | 0.08
Chemical identifiers

| hydrogen fluoride | hydrogen bromide CAS number | 7664-39-3 | 10035-10-6 Beilstein number | | 3587158 PubChem CID number | 16211014 | 260 PubChem SID number | 24863720 | 24857782 SMILES identifier | F | Br InChI identifier | InChI=1/FH/h1H/fF.H/h1h;/q-1;+1 | InChI=1/BrH/h1H RTECS number | MW7875000 | MW3850000 MDL number | MFCD00011346 | MFCD00011323
NFPA label
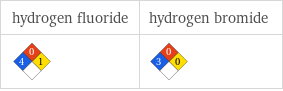
NFPA label

| hydrogen fluoride | hydrogen bromide NFPA health rating | 4 | 3 NFPA fire rating | 0 | 0 NFPA reactivity rating | 1 | 0