Input interpretation
calcium sulfide | tantalum(V) methoxide
Chemical names and formulas
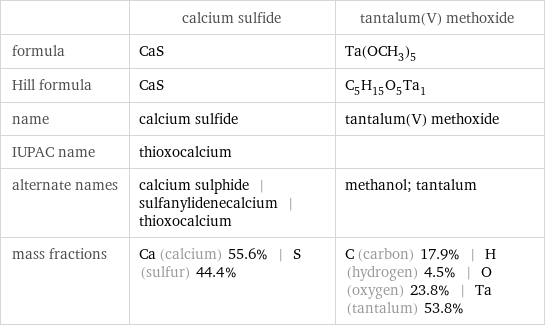
| calcium sulfide | tantalum(V) methoxide formula | CaS | Ta(OCH_3)_5 Hill formula | CaS | C_5H_15O_5Ta_1 name | calcium sulfide | tantalum(V) methoxide IUPAC name | thioxocalcium | alternate names | calcium sulphide | sulfanylidenecalcium | thioxocalcium | methanol; tantalum mass fractions | Ca (calcium) 55.6% | S (sulfur) 44.4% | C (carbon) 17.9% | H (hydrogen) 4.5% | O (oxygen) 23.8% | Ta (tantalum) 53.8%
Structure diagrams

Structure diagrams
Basic properties
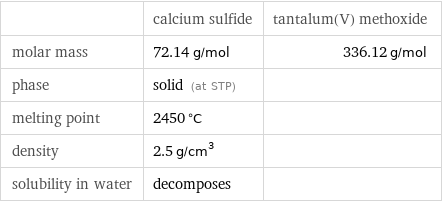
| calcium sulfide | tantalum(V) methoxide molar mass | 72.14 g/mol | 336.12 g/mol phase | solid (at STP) | melting point | 2450 °C | density | 2.5 g/cm^3 | solubility in water | decomposes |
Units
