Input interpretation
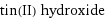
tin(II) hydroxide
Chemical names and formulas

formula | Sn(OH)_2 Hill formula | H_2O_2Sn name | tin(II) hydroxide IUPAC name | stannous dihydroxide alternate names | stannous dihydroxide | tin(+2) cation dihydroxide mass fractions | H (hydrogen) 1.32% | O (oxygen) 21% | Sn (tin) 77.7%
Structure diagram
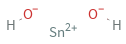
Structure diagram
Basic properties
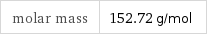
molar mass | 152.72 g/mol
Units

Thermodynamic properties
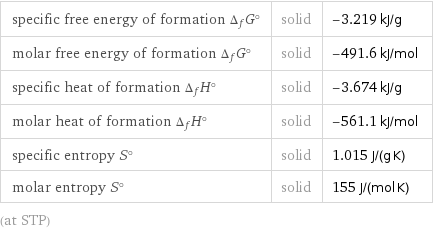
specific free energy of formation Δ_fG° | solid | -3.219 kJ/g molar free energy of formation Δ_fG° | solid | -491.6 kJ/mol specific heat of formation Δ_fH° | solid | -3.674 kJ/g molar heat of formation Δ_fH° | solid | -561.1 kJ/mol specific entropy S° | solid | 1.015 J/(g K) molar entropy S° | solid | 155 J/(mol K) (at STP)
Chemical identifiers
![PubChem CID number | 9793862 SMILES identifier | [OH-].[OH-].[Sn+2] InChI identifier | InChI=1/2H2O.Sn/h2*1H2;/q;;+2/p-2/f2HO.Sn/h2*1h;/q2*-1;m](../image_source/cbb32bbc18d19a5461fb7b76718f6c52.png)
PubChem CID number | 9793862 SMILES identifier | [OH-].[OH-].[Sn+2] InChI identifier | InChI=1/2H2O.Sn/h2*1H2;/q;;+2/p-2/f2HO.Sn/h2*1h;/q2*-1;m
Ion equivalents

Sn^(2+) (tin(II) cation) | 1 (OH)^- (hydroxide anion) | 2