Input interpretation

alkali metals | electron affinity
Summary

median | 48.4 kJ/mol highest | 59.6 kJ/mol (lithium) lowest | 45.5 kJ/mol (cesium) distribution | | (based on 5 values; 1 unavailable)
Entity with missing value
francium
Ranked values

| | visual | ratios | 5 | cesium | | 0.76 | 1 4 | rubidium | | 0.79 | 1.03 3 | potassium | | 0.81 | 1.06 2 | sodium | | 0.89 | 1.16 1 | lithium | | 1 | 1.31
Plot
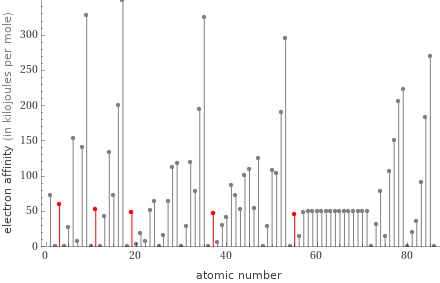
Plot
Periodic table location
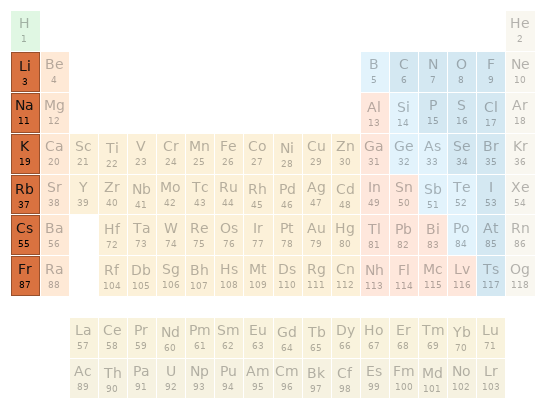
Periodic table location
Electron affinity rankings
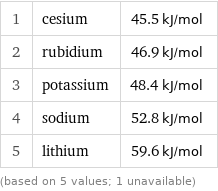
1 | cesium | 45.5 kJ/mol 2 | rubidium | 46.9 kJ/mol 3 | potassium | 48.4 kJ/mol 4 | sodium | 52.8 kJ/mol 5 | lithium | 59.6 kJ/mol (based on 5 values; 1 unavailable)
Unit conversions for median electron affinity 48.4 kJ/mol

0.0484 MJ/mol (megajoules per mole)
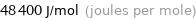
48400 J/mol (joules per mole)

11.6 kcal/mol (thermochemical kilocalories per mole)
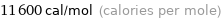
11600 cal/mol (calories per mole)

0.502 eV (molar electronvolts)