Input interpretation
group 6 elements
Members
chromium | molybdenum | tungsten | seaborgium
Periodic table location
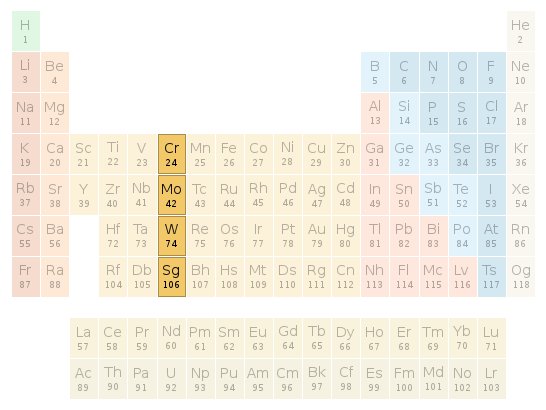
Periodic table location
Images

Images
Basic elemental properties
![| atomic symbol | atomic number | short electronic configuration chromium | Cr | 24 | [Ar]4s^13d^5 molybdenum | Mo | 42 | [Kr]5s^14d^5 tungsten | W | 74 | [Xe]6s^24f^145d^4 seaborgium | Sg | 106 | [Rn]7s^25f^146d^4 | Aufbau diagram | block | group chromium | 3d 4s | d | 6 molybdenum | 4d 5s | d | 6 tungsten | 5d 4f 6s | d | 6 seaborgium | 6d 5f 7s | d | 6 | period | atomic mass | half-life chromium | 4 | 51.9961 u | (stable) molybdenum | 5 | 95.95 u | (stable) tungsten | 6 | 183.84 u | (stable) seaborgium | 7 | 269 u | 120 min](../image_source/fc5220a904717a6a2d96bfa452bab937.png)
| atomic symbol | atomic number | short electronic configuration chromium | Cr | 24 | [Ar]4s^13d^5 molybdenum | Mo | 42 | [Kr]5s^14d^5 tungsten | W | 74 | [Xe]6s^24f^145d^4 seaborgium | Sg | 106 | [Rn]7s^25f^146d^4 | Aufbau diagram | block | group chromium | 3d 4s | d | 6 molybdenum | 4d 5s | d | 6 tungsten | 5d 4f 6s | d | 6 seaborgium | 6d 5f 7s | d | 6 | period | atomic mass | half-life chromium | 4 | 51.9961 u | (stable) molybdenum | 5 | 95.95 u | (stable) tungsten | 6 | 183.84 u | (stable) seaborgium | 7 | 269 u | 120 min
Thermodynamic properties

phase at STP | all | solid melting point | median | 2623 °C | highest | 3422 °C (tungsten) | lowest | 1907 °C (chromium) | distribution | boiling point | median | 4639 °C | highest | 5555 °C (tungsten) | lowest | 2671 °C (chromium) | distribution | molar heat of fusion | median | 35 kJ/mol | highest | 36 kJ/mol (molybdenum) | lowest | 20.5 kJ/mol (chromium) molar heat of vaporization | median | 600 kJ/mol | highest | 800 kJ/mol (tungsten) | lowest | 339 kJ/mol (chromium) | distribution | specific heat at STP | median | 251 J/(kg K) | highest | 448 J/(kg K) (chromium) | lowest | 132 J/(kg K) (tungsten) | distribution | (properties at standard conditions)
Material properties
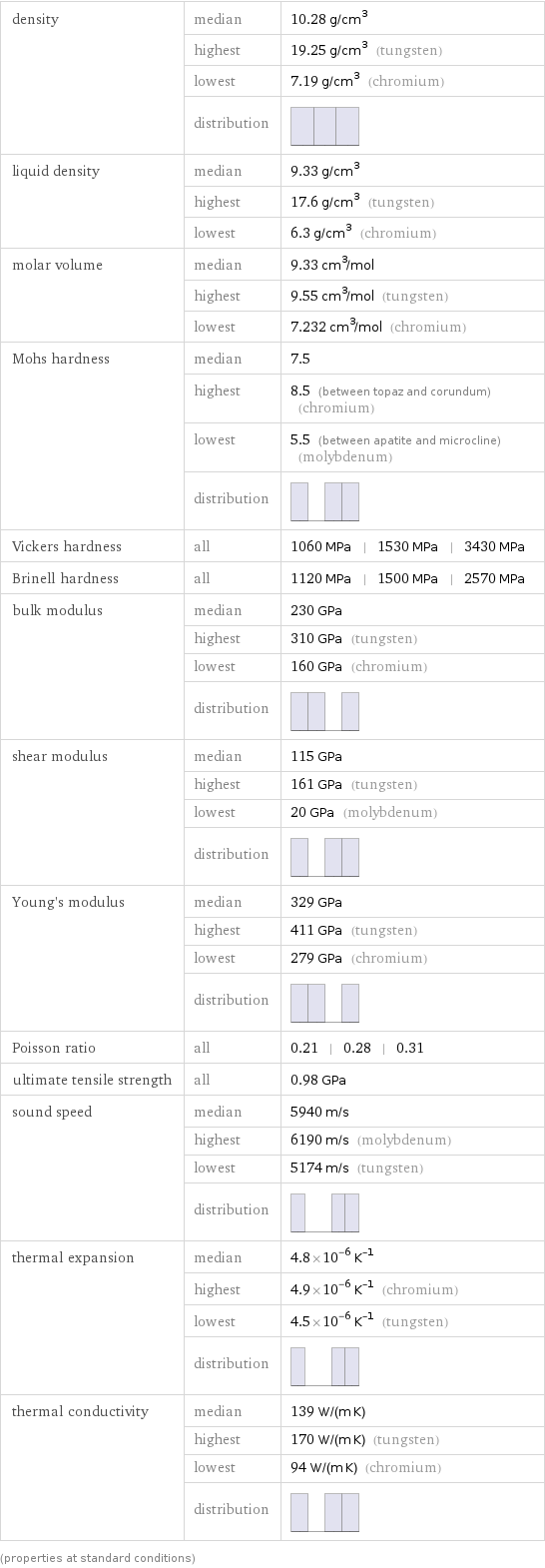
density | median | 10.28 g/cm^3 | highest | 19.25 g/cm^3 (tungsten) | lowest | 7.19 g/cm^3 (chromium) | distribution | liquid density | median | 9.33 g/cm^3 | highest | 17.6 g/cm^3 (tungsten) | lowest | 6.3 g/cm^3 (chromium) molar volume | median | 9.33 cm^3/mol | highest | 9.55 cm^3/mol (tungsten) | lowest | 7.232 cm^3/mol (chromium) Mohs hardness | median | 7.5 | highest | 8.5 (between topaz and corundum) (chromium) | lowest | 5.5 (between apatite and microcline) (molybdenum) | distribution | Vickers hardness | all | 1060 MPa | 1530 MPa | 3430 MPa Brinell hardness | all | 1120 MPa | 1500 MPa | 2570 MPa bulk modulus | median | 230 GPa | highest | 310 GPa (tungsten) | lowest | 160 GPa (chromium) | distribution | shear modulus | median | 115 GPa | highest | 161 GPa (tungsten) | lowest | 20 GPa (molybdenum) | distribution | Young's modulus | median | 329 GPa | highest | 411 GPa (tungsten) | lowest | 279 GPa (chromium) | distribution | Poisson ratio | all | 0.21 | 0.28 | 0.31 ultimate tensile strength | all | 0.98 GPa sound speed | median | 5940 m/s | highest | 6190 m/s (molybdenum) | lowest | 5174 m/s (tungsten) | distribution | thermal expansion | median | 4.8×10^-6 K^(-1) | highest | 4.9×10^-6 K^(-1) (chromium) | lowest | 4.5×10^-6 K^(-1) (tungsten) | distribution | thermal conductivity | median | 139 W/(m K) | highest | 170 W/(m K) (tungsten) | lowest | 94 W/(m K) (chromium) | distribution | (properties at standard conditions)
Electromagnetic properties

resistivity | median | 5×10^-8 Ω m (ohm meters) | highest | 1.3×10^-7 Ω m (ohm meters) (chromium) | lowest | 5×10^-8 Ω m (ohm meters) (molybdenum and tungsten) electrical conductivity | median | 2×10^7 S/m (siemens per meter) | highest | 2×10^7 S/m (siemens per meter) (molybdenum and tungsten) | lowest | 7.9×10^6 S/m (siemens per meter) (chromium) volume magnetic susceptibility | median | 1.203×10^-4 | highest | 3.177×10^-4 (chromium) | lowest | 8.84×10^-5 (tungsten) | distribution | mass magnetic susceptibility | median | 1.17×10^-8 m^3/kg (cubic meters per kilogram) | highest | 4.45×10^-8 m^3/kg (cubic meters per kilogram) (chromium) | lowest | 4.59×10^-9 m^3/kg (cubic meters per kilogram) (tungsten) | distribution | molar magnetic susceptibility | median | 1.122×10^-9 m^3/mol (cubic meters per mole) | highest | 2.314×10^-9 m^3/mol (cubic meters per mole) (chromium) | lowest | 8.44×10^-10 m^3/mol (cubic meters per mole) (tungsten) | distribution | work function | all | 4.5 eV | (4.32 to 5.22) eV | (4.36 to 4.95) eV
Reactivity
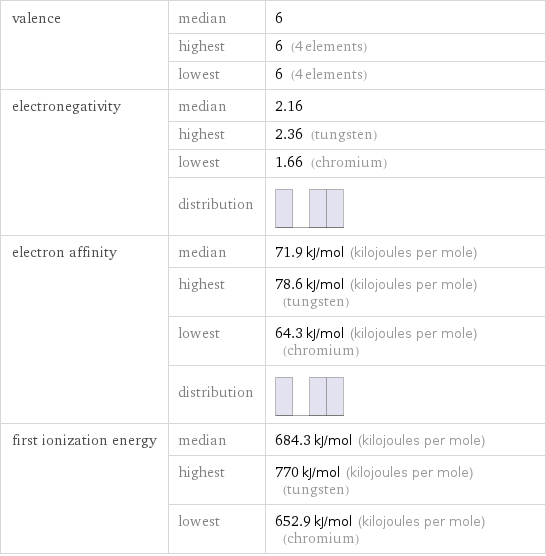
valence | median | 6 | highest | 6 (4 elements) | lowest | 6 (4 elements) electronegativity | median | 2.16 | highest | 2.36 (tungsten) | lowest | 1.66 (chromium) | distribution | electron affinity | median | 71.9 kJ/mol (kilojoules per mole) | highest | 78.6 kJ/mol (kilojoules per mole) (tungsten) | lowest | 64.3 kJ/mol (kilojoules per mole) (chromium) | distribution | first ionization energy | median | 684.3 kJ/mol (kilojoules per mole) | highest | 770 kJ/mol (kilojoules per mole) (tungsten) | lowest | 652.9 kJ/mol (kilojoules per mole) (chromium)
Atomic properties

term symbol | all | ^5D_0 | ^7S_3 atomic radius | median | 140 pm | highest | 145 pm (molybdenum) | lowest | 135 pm (tungsten) | distribution | covalent radius | median | 154 pm | highest | 162 pm (tungsten) | lowest | 139 pm (chromium) | distribution | (electronic ground state properties)