Input interpretation
stannous oxide
Chemical names and formulas

formula | SnO Hill formula | OSn name | stannous oxide alternate names | ketotin | oxotin | tin(II) oxide | tin monoxide | tin oxide mass fractions | O (oxygen) 11.9% | Sn (tin) 88.1%
Structure diagram
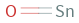
Structure diagram
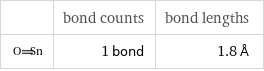
| bond counts | bond lengths | 1 bond | 1.8 Å
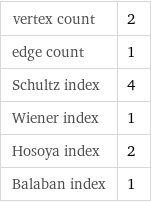
vertex count | 2 edge count | 1 Schultz index | 4 Wiener index | 1 Hosoya index | 2 Balaban index | 1
Basic properties

molar mass | 134.709 g/mol phase | solid (at STP) melting point | 1040 °C boiling point | 1425 °C density | 6.45 g/cm^3 solubility in water | insoluble particle size | 10 µm
Solid properties (at STP)

density | 6.45 g/cm^3
Units

Thermodynamic properties
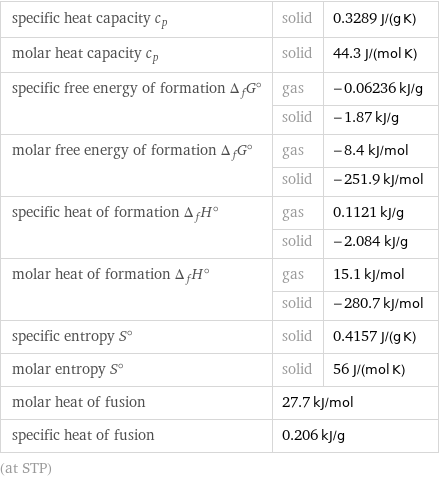
specific heat capacity c_p | solid | 0.3289 J/(g K) molar heat capacity c_p | solid | 44.3 J/(mol K) specific free energy of formation Δ_fG° | gas | -0.06236 kJ/g | solid | -1.87 kJ/g molar free energy of formation Δ_fG° | gas | -8.4 kJ/mol | solid | -251.9 kJ/mol specific heat of formation Δ_fH° | gas | 0.1121 kJ/g | solid | -2.084 kJ/g molar heat of formation Δ_fH° | gas | 15.1 kJ/mol | solid | -280.7 kJ/mol specific entropy S° | solid | 0.4157 J/(g K) molar entropy S° | solid | 56 J/(mol K) molar heat of fusion | 27.7 kJ/mol | specific heat of fusion | 0.206 kJ/g | (at STP)
Chemical identifiers

CAS number | 21651-19-4 RTECS number | XQ3700000 MDL number | MFCD00011243
NFPA label

NFPA label

NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

RTECS classes | other