Input interpretation
potassium iodide | sodium bromide
Chemical names and formulas
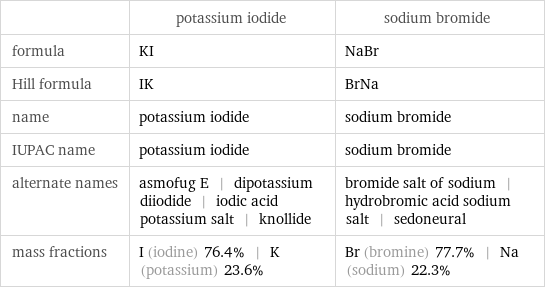
| potassium iodide | sodium bromide formula | KI | NaBr Hill formula | IK | BrNa name | potassium iodide | sodium bromide IUPAC name | potassium iodide | sodium bromide alternate names | asmofug E | dipotassium diiodide | iodic acid potassium salt | knollide | bromide salt of sodium | hydrobromic acid sodium salt | sedoneural mass fractions | I (iodine) 76.4% | K (potassium) 23.6% | Br (bromine) 77.7% | Na (sodium) 22.3%
Structure diagrams

Structure diagrams
Basic properties
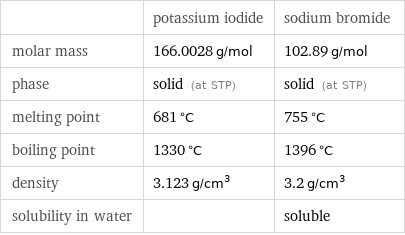
| potassium iodide | sodium bromide molar mass | 166.0028 g/mol | 102.89 g/mol phase | solid (at STP) | solid (at STP) melting point | 681 °C | 755 °C boiling point | 1330 °C | 1396 °C density | 3.123 g/cm^3 | 3.2 g/cm^3 solubility in water | | soluble
Units

Thermodynamic properties

| potassium iodide | sodium bromide specific heat of vaporization | | 1.757 kJ/g (kilojoules per gram) specific heat of fusion | 0.14 kJ/g (kilojoules per gram) | 0.2549 kJ/g (kilojoules per gram)
Solid properties (at STP)
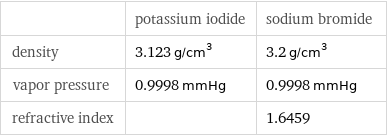
| potassium iodide | sodium bromide density | 3.123 g/cm^3 | 3.2 g/cm^3 vapor pressure | 0.9998 mmHg | 0.9998 mmHg refractive index | | 1.6459
Units

Chemical identifiers
![| potassium iodide | sodium bromide CAS number | 7681-11-0 | 7647-15-6 PubChem CID number | 4875 | 253881 PubChem SID number | 24852091 | 24853756 SMILES identifier | [K+].[I-] | [Na+].[Br-]](../image_source/ee5174c498ab8261665de88104f36733.png)
| potassium iodide | sodium bromide CAS number | 7681-11-0 | 7647-15-6 PubChem CID number | 4875 | 253881 PubChem SID number | 24852091 | 24853756 SMILES identifier | [K+].[I-] | [Na+].[Br-]
NFPA label

NFPA label