Input interpretation
oxoanions
Members

acetate anion | aluminate anion | antimonate anion | antimonite anion | arsenate anion | arsenite anion | beryllate anion | borate anion | bromate anion | bromite anion | carbonate anion | chlorate anion | chlorite anion | chromate anion | chromite anion | cyanate anion | deuteroxide anion | dichromate anion | dihydrogen borate anion | dihydrogen diphosphate anion | dihydrogen hypophosphate anion | dihydrogen phosphate anion | dihydrogen phosphite anion | diphosphate anion | disilicate anion | dithionite anion | ferrate anion | formate anion | hydrogen arsenate anion | hydrogen borate anion | hydrogen carbonate anion | hydrogen chromate anion | hydrogen diphosphate anion | hydrogen molybdate anion | hydrogen phosphate anion | hydrogen phosphite anion | hydrogen selenate anion | hydrogen sulfate anion | hydrogen sulfite anion | hydroperoxide anion | ... (total: 85)
Ionic radii
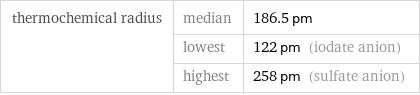
thermochemical radius | median | 186.5 pm | lowest | 122 pm (iodate anion) | highest | 258 pm (sulfate anion)
Units
