Input interpretation
propionaldehyde
Chemical names and formulas
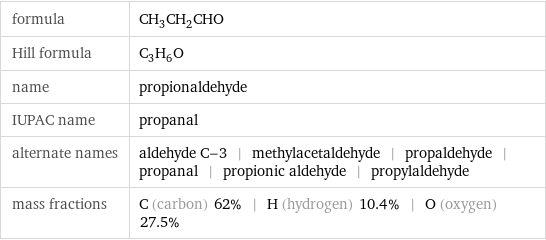
formula | CH_3CH_2CHO Hill formula | C_3H_6O name | propionaldehyde IUPAC name | propanal alternate names | aldehyde C-3 | methylacetaldehyde | propaldehyde | propanal | propionic aldehyde | propylaldehyde mass fractions | C (carbon) 62% | H (hydrogen) 10.4% | O (oxygen) 27.5%
Lewis structure

Draw the Lewis structure of propionaldehyde. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 3 n_C, val + 6 n_H, val + n_O, val = 24 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 3 n_C, full + 6 n_H, full + n_O, full = 44 Subtracting these two numbers shows that 44 - 24 = 20 bonding electrons are needed. Each bond has two electrons, so in addition to the 9 bonds already present in the diagram add 1 bond. To minimize formal charge oxygen wants 2 bonds and carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 1 bond by pairing electrons between adjacent highlighted atoms: Answer: | |
3D structure
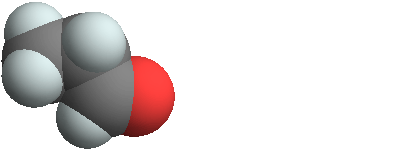
3D structure
Basic properties
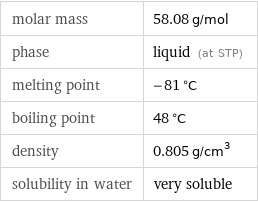
molar mass | 58.08 g/mol phase | liquid (at STP) melting point | -81 °C boiling point | 48 °C density | 0.805 g/cm^3 solubility in water | very soluble
Units

Liquid properties (at STP)
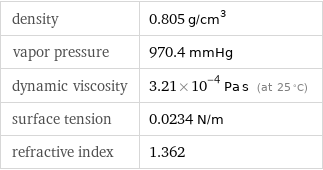
density | 0.805 g/cm^3 vapor pressure | 970.4 mmHg dynamic viscosity | 3.21×10^-4 Pa s (at 25 °C) surface tension | 0.0234 N/m refractive index | 1.362
Units
Thermodynamic properties
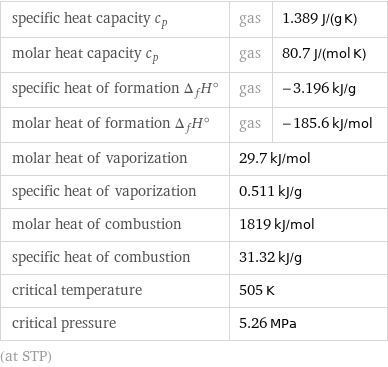
specific heat capacity c_p | gas | 1.389 J/(g K) molar heat capacity c_p | gas | 80.7 J/(mol K) specific heat of formation Δ_fH° | gas | -3.196 kJ/g molar heat of formation Δ_fH° | gas | -185.6 kJ/mol molar heat of vaporization | 29.7 kJ/mol | specific heat of vaporization | 0.511 kJ/g | molar heat of combustion | 1819 kJ/mol | specific heat of combustion | 31.32 kJ/g | critical temperature | 505 K | critical pressure | 5.26 MPa | (at STP)
Chemical identifiers
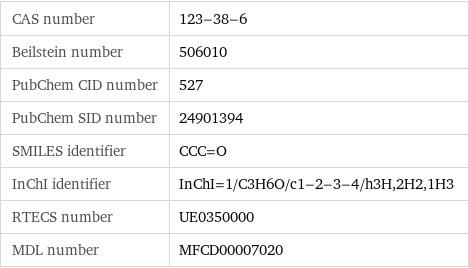
CAS number | 123-38-6 Beilstein number | 506010 PubChem CID number | 527 PubChem SID number | 24901394 SMILES identifier | CCC=O InChI identifier | InChI=1/C3H6O/c1-2-3-4/h3H, 2H2, 1H3 RTECS number | UE0350000 MDL number | MFCD00007020
NFPA label

NFPA label

NFPA health rating | 2 NFPA fire rating | 3 NFPA reactivity rating | 2
Safety properties
flash point | -26.67 °C autoignition point | 207 °C lower explosive limit | 2.9% (concentration in air) upper explosive limit | 17% (concentration in air)

DOT hazard class | 3 DOT numbers | 1275
Toxicity properties

threshold limit value | 20 ppmv

RTECS classes | mutagen | primary irritant