Input interpretation
chlorine (chemical element) | nitrogen (chemical element) | krypton (chemical element)
Periodic table location
Periodic table location
Images

Images
Basic elemental properties
![| chlorine | nitrogen | krypton atomic symbol | Cl | N | Kr atomic number | 17 | 7 | 36 short electronic configuration | [Ne]3s^23p^5 | [He]2s^22p^3 | [Ar]4s^23d^104p^6 Aufbau diagram | 3p 3s | 2p 2s | 4p 3d 4s block | p | p | p group | 17 | 15 | 18 period | 3 | 2 | 4 atomic mass | 35.45 u | 14.007 u | 83.798 u half-life | (stable) | (stable) | (stable)](../image_source/cc4c3b2b8d6f2350dc05b304e6179f4d.png)
| chlorine | nitrogen | krypton atomic symbol | Cl | N | Kr atomic number | 17 | 7 | 36 short electronic configuration | [Ne]3s^23p^5 | [He]2s^22p^3 | [Ar]4s^23d^104p^6 Aufbau diagram | 3p 3s | 2p 2s | 4p 3d 4s block | p | p | p group | 17 | 15 | 18 period | 3 | 2 | 4 atomic mass | 35.45 u | 14.007 u | 83.798 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
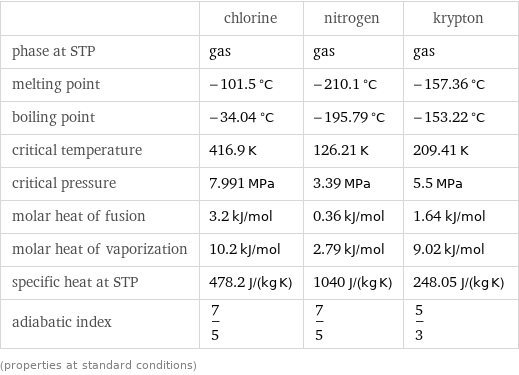
| chlorine | nitrogen | krypton phase at STP | gas | gas | gas melting point | -101.5 °C | -210.1 °C | -157.36 °C boiling point | -34.04 °C | -195.79 °C | -153.22 °C critical temperature | 416.9 K | 126.21 K | 209.41 K critical pressure | 7.991 MPa | 3.39 MPa | 5.5 MPa molar heat of fusion | 3.2 kJ/mol | 0.36 kJ/mol | 1.64 kJ/mol molar heat of vaporization | 10.2 kJ/mol | 2.79 kJ/mol | 9.02 kJ/mol specific heat at STP | 478.2 J/(kg K) | 1040 J/(kg K) | 248.05 J/(kg K) adiabatic index | 7/5 | 7/5 | 5/3 (properties at standard conditions)
Material properties
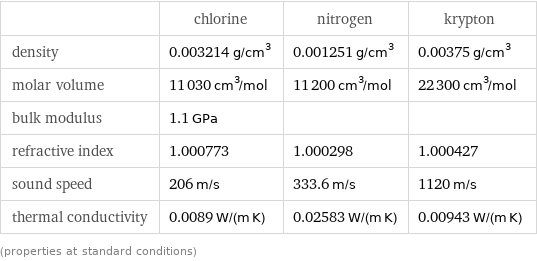
| chlorine | nitrogen | krypton density | 0.003214 g/cm^3 | 0.001251 g/cm^3 | 0.00375 g/cm^3 molar volume | 11030 cm^3/mol | 11200 cm^3/mol | 22300 cm^3/mol bulk modulus | 1.1 GPa | | refractive index | 1.000773 | 1.000298 | 1.000427 sound speed | 206 m/s | 333.6 m/s | 1120 m/s thermal conductivity | 0.0089 W/(m K) | 0.02583 W/(m K) | 0.00943 W/(m K) (properties at standard conditions)
Electromagnetic properties
| chlorine | nitrogen | krypton electrical type | insulator | | resistivity | 100 Ω m (ohm meters) | | electrical conductivity | 0.01 S/m (siemens per meter) | | magnetic type | diamagnetic | diamagnetic | diamagnetic volume magnetic susceptibility | -2.31×10^-8 | -6.8×10^-9 | -1.65×10^-8 mass magnetic susceptibility | -7.2×10^-9 m^3/kg (cubic meters per kilogram) | -5.4×10^-9 m^3/kg (cubic meters per kilogram) | -4.4×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -5.11×10^-10 m^3/mol (cubic meters per mole) | -1.5×10^-10 m^3/mol (cubic meters per mole) | -3.69×10^-10 m^3/mol (cubic meters per mole) color | (yellow) | (colorless) | (colorless) refractive index | 1.000773 | 1.000298 | 1.000427
Reactivity
| chlorine | nitrogen | krypton valence | 5 | 3 | 2 electronegativity | 3.16 | 3.04 | 3 electron affinity | 349 kJ/mol (kilojoules per mole) | 7 kJ/mol (kilojoules per mole) | 0 kJ/mol (kilojoules per mole) first ionization energy | 1251.2 kJ/mol (kilojoules per mole) | 1402.3 kJ/mol (kilojoules per mole) | 1350.8 kJ/mol (kilojoules per mole) ionization energies | 1251.2 kJ/mol | 2298 kJ/mol | 3822 kJ/mol | 5158.6 kJ/mol | 6542 kJ/mol | 9362 kJ/mol | 11018 kJ/mol | 33604 kJ/mol | 38600 kJ/mol | 43961 kJ/mol | 1402.3 kJ/mol | 2856 kJ/mol | 4578.1 kJ/mol | 7475 kJ/mol | 9444.9 kJ/mol | 53266.6 kJ/mol | 64360 kJ/mol | 1350.8 kJ/mol | 2350.4 kJ/mol | 3565 kJ/mol | 5070 kJ/mol | 6240 kJ/mol | 7570 kJ/mol | 10710 kJ/mol | 12138 kJ/mol | 22274 kJ/mol | 25880 kJ/mol | 29700 kJ/mol | 33800 kJ/mol | 37700 kJ/mol | 43100 kJ/mol | 47500 kJ/mol | 52200 kJ/mol | 57100 kJ/mol | 61800 kJ/mol | 75800 kJ/mol | 80400 kJ/mol | 85300 kJ/mol
Reactivity
Atomic properties

| chlorine | nitrogen | krypton term symbol | ^2P_(3/2) | ^4S_(3/2) | ^1S_0 atomic radius | 100 pm | 65 pm | 88 pm covalent radius | 102 pm | 71 pm | 116 pm van der Waals radius | 175 pm | 155 pm | 202 pm (electronic ground state properties)
Abundances
| chlorine | nitrogen | krypton universe abundance | 1×10^-4 mass% | 0.1 mass% | 4×10^-6 mass% solar abundance | 8×10^-4 mass% | 0.1 mass% | meteorite abundance | 0.037 mass% | 0.14 mass% | crust abundance | 0.017 mass% | 0.002 mass% | 1.5×10^-8 mass% ocean abundance | 2 mass% | 5×10^-5 mass% | 2.1×10^-8 mass% human abundance | 0.12 mass% | 2.6 mass% | universe molar abundance | 4×10^-6 mol% | 0.009 mol% | 6×10^-8 mol% solar molar abundance | 3×10^-5 mol% | 0.009 mol% | meteorite molar abundance | 0.016 mol% | 0.14 mol% | ocean molar abundance | 0.35 mol% | 2.2×10^-5 mol% | 1.6×10^-9 mol% crust molar abundance | 0.0099 mol% | 0.0029 mol% | 4×10^-9 mol% human molar abundance | 0.021 mol% | 1.2 mol% |
Nuclear properties
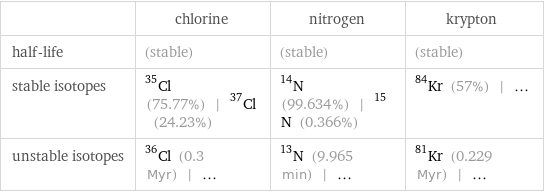
| chlorine | nitrogen | krypton half-life | (stable) | (stable) | (stable) stable isotopes | Cl-35 (75.77%) | Cl-37 (24.23%) | N-14 (99.634%) | N-15 (0.366%) | Kr-84 (57%) | ... unstable isotopes | Cl-36 (0.3 Myr) | ... | N-13 (9.965 min) | ... | Kr-81 (0.229 Myr) | ...
Units

Identifiers
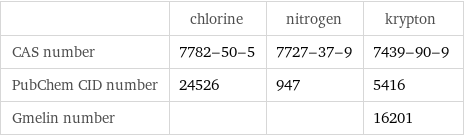
| chlorine | nitrogen | krypton CAS number | 7782-50-5 | 7727-37-9 | 7439-90-9 PubChem CID number | 24526 | 947 | 5416 Gmelin number | | | 16201