Input interpretation
rhodium(I) cation | tetrafluoroborate anion
Structure diagrams
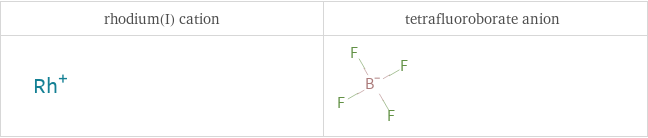
Structure diagrams
General properties
![| rhodium(I) cation | tetrafluoroborate anion formula | Rh^+ | ([BF_4])^- net ionic charge | +1 | -1 alternate names | rhodium(I) | rhodium(1+) | tetrafluoroborate | tetrafluoroborate(1-) | tetrafluoroborate(III)](../image_source/1b126236a78455739582a697d73cfdca.png)
| rhodium(I) cation | tetrafluoroborate anion formula | Rh^+ | ([BF_4])^- net ionic charge | +1 | -1 alternate names | rhodium(I) | rhodium(1+) | tetrafluoroborate | tetrafluoroborate(1-) | tetrafluoroborate(III)
Ionic radii

| tetrafluoroborate anion thermochemical radius | 232 pm
Units

Other properties

| rhodium(I) cation | tetrafluoroborate anion ion class | cations | d block ions | group 9 ions | transition metal ions | anions | nonmetal halide ion | polyatomic ions