Input interpretation
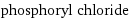
phosphoryl chloride
Chemical names and formulas

formula | POCl_3 Hill formula | Cl_3OP name | phosphoryl chloride alternate names | phosphorus oxychloride | phosphorus oxytrichloride | phosphorus(V) oxide chloride | phosphoryl trichloride | trichlorophosphine oxide | trichlorophosphorus oxide mass fractions | Cl (chlorine) 69.4% | O (oxygen) 10.4% | P (phosphorus) 20.2%
Lewis structure

Draw the Lewis structure of phosphoryl chloride. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the chlorine (n_Cl, val = 7), oxygen (n_O, val = 6), and phosphorus (n_P, val = 5) atoms: 3 n_Cl, val + n_O, val + n_P, val = 32 Calculate the number of electrons needed to completely fill the valence shells for chlorine (n_Cl, full = 8), oxygen (n_O, full = 8), and phosphorus (n_P, full = 8): 3 n_Cl, full + n_O, full + n_P, full = 40 Subtracting these two numbers shows that 40 - 32 = 8 bonding electrons are needed. Each bond has two electrons, so we expect that the above diagram has all the necessary bonds. However, to minimize formal charge oxygen wants 2 bonds. Identify the atom that wants additional bonds and the number of electrons remaining on each atom: In order to minimize their formal charge, atoms with large electronegativities can force atoms with smaller electronegativities on period 3 or higher to expand their valence shells. The electronegativities of the atoms are 2.19 (phosphorus), 3.16 (chlorine), and 3.44 (oxygen). Because the electronegativity of phosphorus is smaller than the electronegativity of oxygen, expand the valence shell of phosphorus to 5 bonds. Therefore we add a total of 1 bond to the diagram: Answer: | |
3D structure

3D structure
Basic properties

molar mass | 153.3 g/mol phase | liquid (at STP) melting point | 1.25 °C boiling point | 105.8 °C density | 1.645 g/cm^3 solubility in water | reacts
Units

Liquid properties (at STP)

density | 1.645 g/cm^3 vapor pressure | 104 mmHg
Units

Thermodynamic properties
molar heat of vaporization | 34.6 kJ/mol specific heat of vaporization | 0.226 kJ/g molar heat of fusion | 13.1 kJ/mol specific heat of fusion | 0.0854 kJ/g critical temperature | 605 K (at STP)
Chemical identifiers
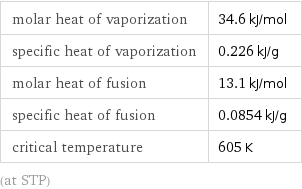
CAS number | 10025-87-3 PubChem CID number | 24813 PubChem SID number | 24855857 SMILES identifier | O=P(Cl)(Cl)Cl InChI identifier | InChI=1/Cl3OP/c1-5(2, 3)4 RTECS number | TH4897000 MDL number | MFCD00011443
NFPA label
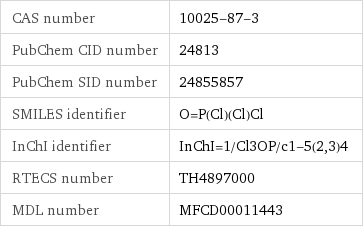
NFPA label
NFPA health rating | 4 NFPA fire rating | 0 NFPA reactivity rating | 2 NFPA hazards | water reactive
Safety properties

flash point | -18 °C
DOT hazard class | 8 DOT numbers | 1810
Toxicity properties
lethal dosage | 330 mg/kg (oral dose for rats) short-term exposure limit | 3.6 mg/m^3 threshold limit value | 0.1 ppmv
probable lethal dose for man | 30 mL (milliliters) long-term exposure limit | 1.2 mg/m^3 (over 8 hours) RTECS classes | other