Input interpretation
hypobromite anion | hydride anion | phosphite anion
Structure diagrams
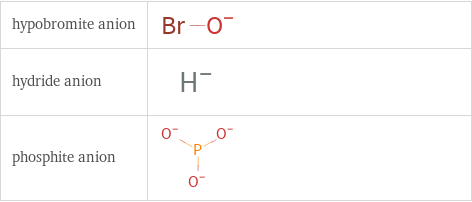
Structure diagrams
General properties
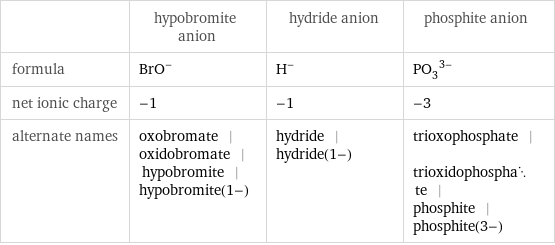
| hypobromite anion | hydride anion | phosphite anion formula | (BrO)^- | H^- | (PO_3)^(3-) net ionic charge | -1 | -1 | -3 alternate names | oxobromate | oxidobromate | hypobromite | hypobromite(1-) | hydride | hydride(1-) | trioxophosphate | trioxidophosphate | phosphite | phosphite(3-)
Other properties
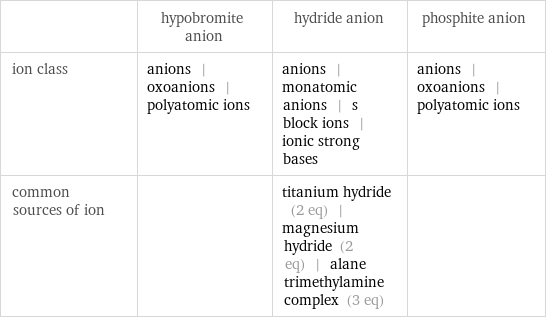
| hypobromite anion | hydride anion | phosphite anion ion class | anions | oxoanions | polyatomic ions | anions | monatomic anions | s block ions | ionic strong bases | anions | oxoanions | polyatomic ions common sources of ion | | titanium hydride (2 eq) | magnesium hydride (2 eq) | alane trimethylamine complex (3 eq) |