Input interpretation
iron(III) oxide | potassium tellurate(VI) trihydrate
Chemical names and formulas
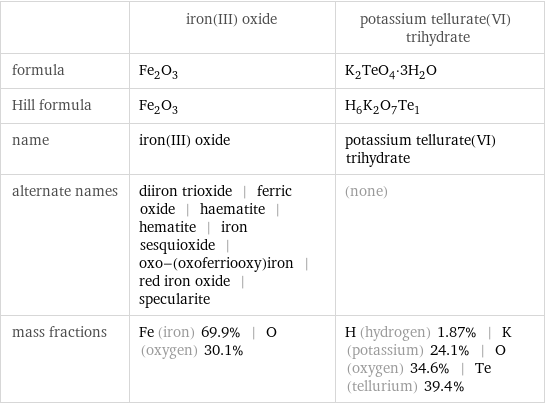
| iron(III) oxide | potassium tellurate(VI) trihydrate formula | Fe_2O_3 | K_2TeO_4·3H_2O Hill formula | Fe_2O_3 | H_6K_2O_7Te_1 name | iron(III) oxide | potassium tellurate(VI) trihydrate alternate names | diiron trioxide | ferric oxide | haematite | hematite | iron sesquioxide | oxo-(oxoferriooxy)iron | red iron oxide | specularite | (none) mass fractions | Fe (iron) 69.9% | O (oxygen) 30.1% | H (hydrogen) 1.87% | K (potassium) 24.1% | O (oxygen) 34.6% | Te (tellurium) 39.4%
Structure diagrams

Structure diagrams
Basic properties
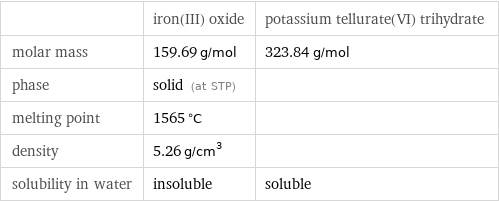
| iron(III) oxide | potassium tellurate(VI) trihydrate molar mass | 159.69 g/mol | 323.84 g/mol phase | solid (at STP) | melting point | 1565 °C | density | 5.26 g/cm^3 | solubility in water | insoluble | soluble
Units
