Input interpretation
diborane
Chemical names and formulas

formula | B_2H_6 name | diborane alternate names | boroethane | boron hydride | diboron hexahydride mass fractions | B (boron) 78.1% | H (hydrogen) 21.9%
Structure diagram
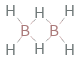
Structure diagram

| bond counts | bond energies | 8 bonds | 389 kJ/mol
3D structure
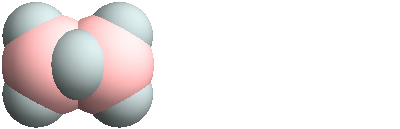
3D structure
Basic properties
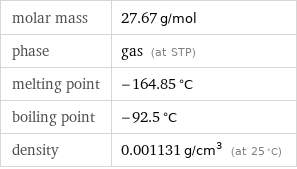
molar mass | 27.67 g/mol phase | gas (at STP) melting point | -164.85 °C boiling point | -92.5 °C density | 0.001131 g/cm^3 (at 25 °C)
Units

Gas properties (at STP)

density | 0.001131 g/cm^3 (at 25 °C) molar volume | 24460 cm^3/mol
Units

Thermodynamic properties

specific heat capacity c_p | gas | 2.049 J/(g K) molar heat capacity c_p | gas | 56.7 J/(mol K) specific free energy of formation Δ_fG° | gas | 3.166 kJ/g | gas | 27.97 kJ/g molar free energy of formation Δ_fG° | gas | 87.6 kJ/mol | gas | 774 kJ/mol specific heat of formation Δ_fH° | gas | 1.316 kJ/g molar heat of formation Δ_fH° | gas | 36.4 kJ/mol critical temperature | 289.8 K | critical pressure | 4.05 MPa | (at STP)
Chemical identifiers
![CAS number | 19287-45-7 SMILES identifier | [H][B]1([H])[H][B]([H])([H])[H]1 InChI identifier | InChI=1S/B2H6/c1-3-2-4-1/h1-2H2 MDL number | MFCD01310566](../image_source/ec992867710f48b3ced10b10de62a607.png)
CAS number | 19287-45-7 SMILES identifier | [H][B]1([H])[H][B]([H])([H])[H]1 InChI identifier | InChI=1S/B2H6/c1-3-2-4-1/h1-2H2 MDL number | MFCD01310566
Toxicity properties

long-term exposure limit | 0.1 mg/m^3 (over 8 hours)
Units
