Input interpretation

mineral acids | Hill formula string
Table
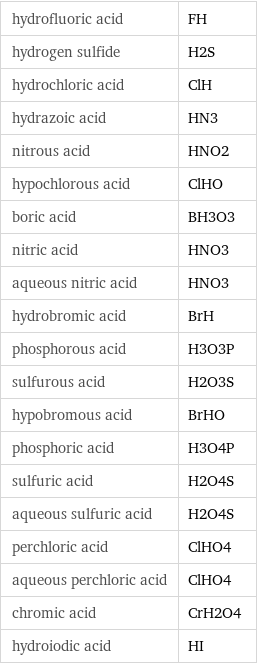
hydrofluoric acid | FH hydrogen sulfide | H2S hydrochloric acid | ClH hydrazoic acid | HN3 nitrous acid | HNO2 hypochlorous acid | ClHO boric acid | BH3O3 nitric acid | HNO3 aqueous nitric acid | HNO3 hydrobromic acid | BrH phosphorous acid | H3O3P sulfurous acid | H2O3S hypobromous acid | BrHO phosphoric acid | H3O4P sulfuric acid | H2O4S aqueous sulfuric acid | H2O4S perchloric acid | ClHO4 aqueous perchloric acid | ClHO4 chromic acid | CrH2O4 hydroiodic acid | HI