Input interpretation

metallic elements | molar heat capacity
Summary

median | 25.7 J/(mol K) highest | 37.7 J/(mol K) (gadolinium) lowest | 6.111 J/(mol K) (technetium) distribution | | (based on 67 values; 27 unavailable)
Entities with missing values

promethium | polonium | francium | neptunium | plutonium | americium | curium | berkelium | californium | einsteinium | ... (total: 27)
Distribution plots
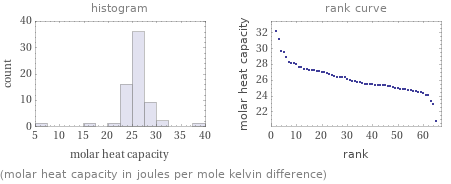
(molar heat capacity in joules per mole kelvin difference)
Molar heat capacity rankings
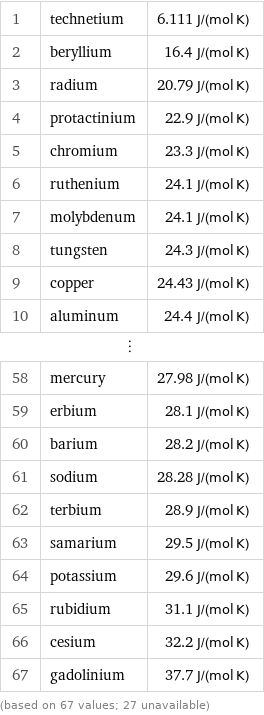
1 | technetium | 6.111 J/(mol K) 2 | beryllium | 16.4 J/(mol K) 3 | radium | 20.79 J/(mol K) 4 | protactinium | 22.9 J/(mol K) 5 | chromium | 23.3 J/(mol K) 6 | ruthenium | 24.1 J/(mol K) 7 | molybdenum | 24.1 J/(mol K) 8 | tungsten | 24.3 J/(mol K) 9 | copper | 24.43 J/(mol K) 10 | aluminum | 24.4 J/(mol K) ⋮ | | 58 | mercury | 27.98 J/(mol K) 59 | erbium | 28.1 J/(mol K) 60 | barium | 28.2 J/(mol K) 61 | sodium | 28.28 J/(mol K) 62 | terbium | 28.9 J/(mol K) 63 | samarium | 29.5 J/(mol K) 64 | potassium | 29.6 J/(mol K) 65 | rubidium | 31.1 J/(mol K) 66 | cesium | 32.2 J/(mol K) 67 | gadolinium | 37.7 J/(mol K) (based on 67 values; 27 unavailable)
Unit conversions for median molar heat capacity 25.7 J/(mol K)
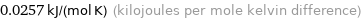
0.0257 kJ/(mol K) (kilojoules per mole kelvin difference)
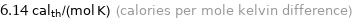
6.14 cal_th/(mol K) (calories per mole kelvin difference)
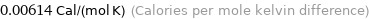
0.00614 Cal/(mol K) (Calories per mole kelvin difference)