Input interpretation
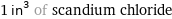
1 in^3 of scandium chloride
Basic properties for 1 in^3
mass | 39.2 grams 0.0392 kg (kilograms) 1.38 oz (ounces) molar amount | 0.259 mol (moles) 259 mmol (millimoles) volume | 16.4 mL (milliliters) 0.0164 L (liters) 16.4 cm^3 (cubic centimeters) 1.64×10^-5 m^3 (cubic meters) 0.554 fl oz (fluid ounces) 3.32 tsp (teaspoons) 1.11 tbsp (tablespoons) 0.0173 quarts equivalents | 0.259 eq (equivalents) (at STP)
Corresponding quantities
sphere radius | 1.576 cm (centimeters) side of a cube | 2.54 cm (centimeters)
Thermodynamic properties for 1 in^3
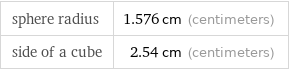
heat of formation Δ_fH° | solid | -239.5 kJ latent heat of fusion | 17.4 kJ (kilojoules) |
Units

Phase change energies for 1 in^3 from 25 °C

energy required to convert to liquid | 17.4 kJ (kilojoules)
Mass composition for 1 in^3
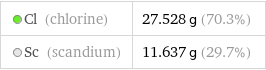
Cl (chlorine) | 27.528 g (70.3%) Sc (scandium) | 11.637 g (29.7%)
Mass composition for 1 in^3
Structure diagram
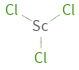
Structure diagram

| bond counts | bond lengths | 3 bonds | 2.2 Å
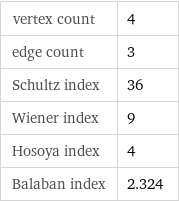
vertex count | 4 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Chemical names and formulas
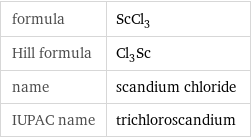
formula | ScCl_3 Hill formula | Cl_3Sc name | scandium chloride IUPAC name | trichloroscandium
Substance properties
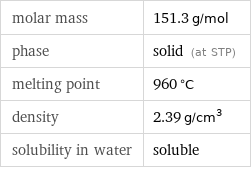
molar mass | 151.3 g/mol phase | solid (at STP) melting point | 960 °C density | 2.39 g/cm^3 solubility in water | soluble
Units
