Input interpretation

dying low mass star elements | atomic radius
Summary
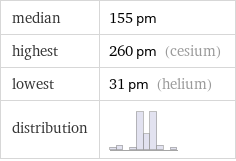
median | 155 pm highest | 260 pm (cesium) lowest | 31 pm (helium) distribution |
Units

Plot
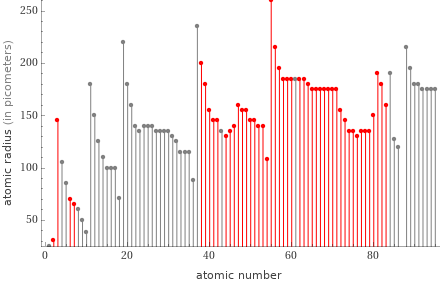
Plot
Periodic table location
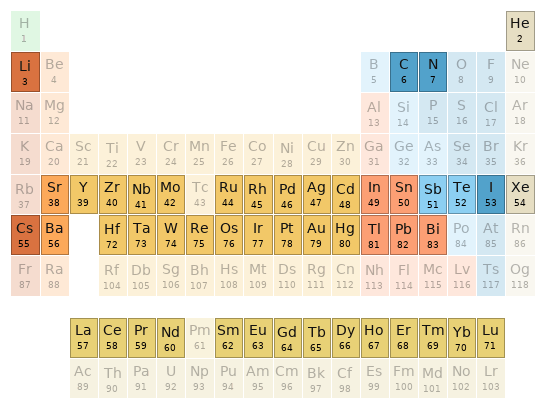
Periodic table location
Atomic radii properties

atomic radius (empirical) | median | 155 pm | highest | 260 pm (cesium) | lowest | 65 pm (nitrogen) | distribution | atomic radius (calculated) | median | 186 pm | highest | 298 pm (cesium) | lowest | 31 pm (helium) | distribution | covalent radius | median | 152 pm | highest | 244 pm (cesium) | lowest | 28 pm (helium) | distribution | van der Waals radius | median | 175 pm | highest | 217 pm (tin) | lowest | 140 pm (helium) | distribution |
Units

Distribution plots
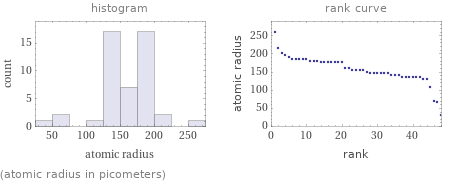
(atomic radius in picometers)
Atomic radius rankings
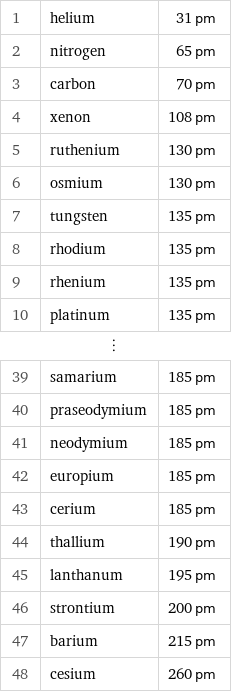
1 | helium | 31 pm 2 | nitrogen | 65 pm 3 | carbon | 70 pm 4 | xenon | 108 pm 5 | ruthenium | 130 pm 6 | osmium | 130 pm 7 | tungsten | 135 pm 8 | rhodium | 135 pm 9 | rhenium | 135 pm 10 | platinum | 135 pm ⋮ | | 39 | samarium | 185 pm 40 | praseodymium | 185 pm 41 | neodymium | 185 pm 42 | europium | 185 pm 43 | cerium | 185 pm 44 | thallium | 190 pm 45 | lanthanum | 195 pm 46 | strontium | 200 pm 47 | barium | 215 pm 48 | cesium | 260 pm
Unit conversions for median atomic radius 155 pm
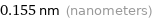
0.155 nm (nanometers)
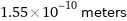
1.55×10^-10 meters
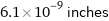
6.1×10^-9 inches
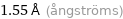
1.55 Å (ångströms)
Comparisons for median atomic radius 155 pm
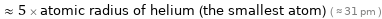
≈ 5 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
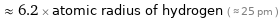
≈ 6.2 × atomic radius of hydrogen ( ≈ 25 pm )