Input interpretation

actinide ions | molar mass
Summary
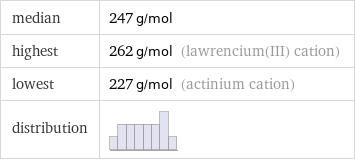
median | 247 g/mol highest | 262 g/mol (lawrencium(III) cation) lowest | 227 g/mol (actinium cation) distribution |
Units

Distribution plots
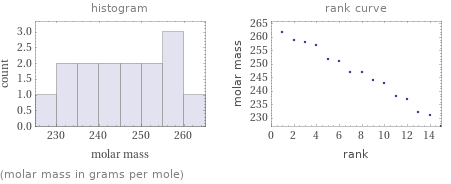
(molar mass in grams per mole)
Molar mass rankings
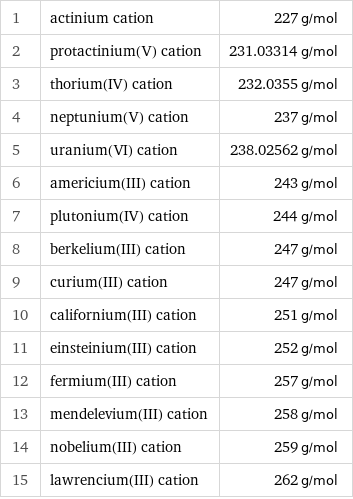
1 | actinium cation | 227 g/mol 2 | protactinium(V) cation | 231.03314 g/mol 3 | thorium(IV) cation | 232.0355 g/mol 4 | neptunium(V) cation | 237 g/mol 5 | uranium(VI) cation | 238.02562 g/mol 6 | americium(III) cation | 243 g/mol 7 | plutonium(IV) cation | 244 g/mol 8 | berkelium(III) cation | 247 g/mol 9 | curium(III) cation | 247 g/mol 10 | californium(III) cation | 251 g/mol 11 | einsteinium(III) cation | 252 g/mol 12 | fermium(III) cation | 257 g/mol 13 | mendelevium(III) cation | 258 g/mol 14 | nobelium(III) cation | 259 g/mol 15 | lawrencium(III) cation | 262 g/mol
Unit conversion for median molar mass 247 g/mol
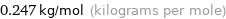
0.247 kg/mol (kilograms per mole)
Comparisons for median molar mass 247 g/mol
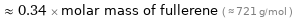
≈ 0.34 × molar mass of fullerene ( ≈ 721 g/mol )
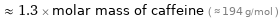
≈ 1.3 × molar mass of caffeine ( ≈ 194 g/mol )
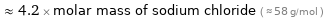
≈ 4.2 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities
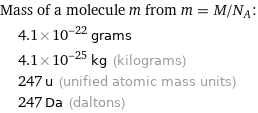
Mass of a molecule m from m = M/N_A: | 4.1×10^-22 grams | 4.1×10^-25 kg (kilograms) | 247 u (unified atomic mass units) | 247 Da (daltons)
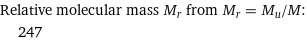
Relative molecular mass M_r from M_r = M_u/M: | 247