Input interpretation
strontium phosphide | lanthanum(III) chloride hydrate
Chemical names and formulas

| strontium phosphide | lanthanum(III) chloride hydrate formula | Sr_3P_2 | LaCl_3·xH_2O Hill formula | P_2Sr_3 | Cl_3H_2LaO name | strontium phosphide | lanthanum(III) chloride hydrate IUPAC name | phosphorus(-3) anion; strontium(+2) cation | trichlorolanthanum hydrate alternate names | strontium phosphide (sr3P2) | tristrontium diphosphide | trichlorolanthanum hydrate mass fractions | P (phosphorus) 19.1% | Sr (strontium) 80.9% | Cl (chlorine) 40.4% | H (hydrogen) 0.766% | La (lanthanum) 52.8% | O (oxygen) 6.08%
Structure diagrams
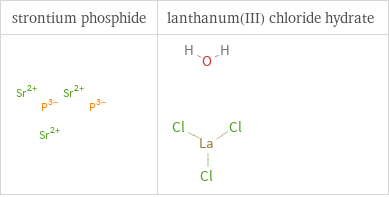
Structure diagrams
Basic properties

| strontium phosphide | lanthanum(III) chloride hydrate molar mass | 324.8 g/mol | 263.3 g/mol phase | | solid (at STP) melting point | | 92.5 °C density | 2.68 g/cm^3 |
Units
