Input interpretation
vanadyl chloride | potassium hexafluoronickelate(IV)
Chemical names and formulas
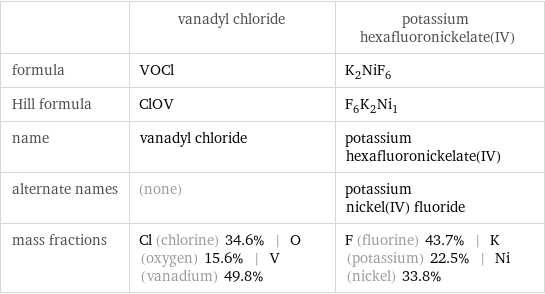
| vanadyl chloride | potassium hexafluoronickelate(IV) formula | VOCl | K_2NiF_6 Hill formula | ClOV | F_6K_2Ni_1 name | vanadyl chloride | potassium hexafluoronickelate(IV) alternate names | (none) | potassium nickel(IV) fluoride mass fractions | Cl (chlorine) 34.6% | O (oxygen) 15.6% | V (vanadium) 49.8% | F (fluorine) 43.7% | K (potassium) 22.5% | Ni (nickel) 33.8%
Structure diagrams
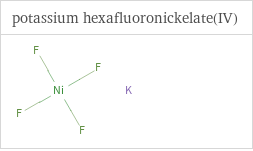
Structure diagrams
Basic properties
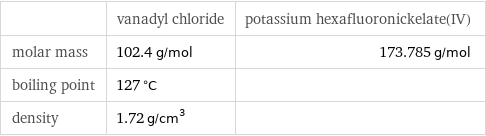
| vanadyl chloride | potassium hexafluoronickelate(IV) molar mass | 102.4 g/mol | 173.785 g/mol boiling point | 127 °C | density | 1.72 g/cm^3 |
Units
