Input interpretation
osmium | thallium(I) fluoride
Chemical names and formulas
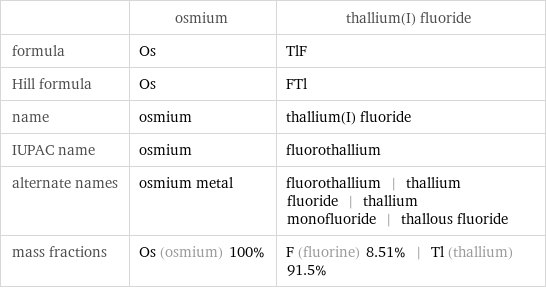
| osmium | thallium(I) fluoride formula | Os | TlF Hill formula | Os | FTl name | osmium | thallium(I) fluoride IUPAC name | osmium | fluorothallium alternate names | osmium metal | fluorothallium | thallium fluoride | thallium monofluoride | thallous fluoride mass fractions | Os (osmium) 100% | F (fluorine) 8.51% | Tl (thallium) 91.5%
Structure diagrams

Structure diagrams
Basic properties
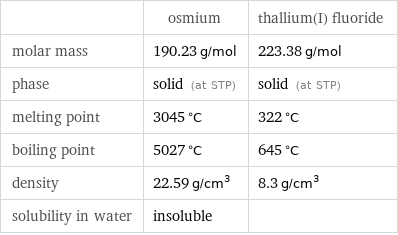
| osmium | thallium(I) fluoride molar mass | 190.23 g/mol | 223.38 g/mol phase | solid (at STP) | solid (at STP) melting point | 3045 °C | 322 °C boiling point | 5027 °C | 645 °C density | 22.59 g/cm^3 | 8.3 g/cm^3 solubility in water | insoluble |
Units

Thermodynamic properties
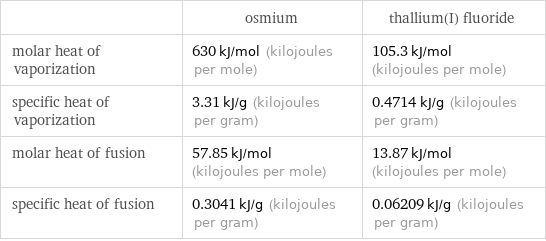
| osmium | thallium(I) fluoride molar heat of vaporization | 630 kJ/mol (kilojoules per mole) | 105.3 kJ/mol (kilojoules per mole) specific heat of vaporization | 3.31 kJ/g (kilojoules per gram) | 0.4714 kJ/g (kilojoules per gram) molar heat of fusion | 57.85 kJ/mol (kilojoules per mole) | 13.87 kJ/mol (kilojoules per mole) specific heat of fusion | 0.3041 kJ/g (kilojoules per gram) | 0.06209 kJ/g (kilojoules per gram)
Solid properties (at STP)

| osmium | thallium(I) fluoride density | 22.59 g/cm^3 | 8.3 g/cm^3
Units
