Input interpretation
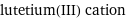
lutetium(III) cation
Structure diagram

Structure diagram
General properties

formula | Lu^(3+) net ionic charge | +3 alternate names | lutetium(III) | lutetium(3+)
Ionic radii

lutetium(III) cation state | radius 6-coordinate | 86 pm 8-coordinate | 97 pm
Units

Other properties

ion class | cations | d block ions | f block ions | group 3 ions | lanthanide ions | lutetium 3 ion | transition metal ions
Thermodynamic properties

molar heat capacity c_p | aqueous | 25 J/(mol K) (joules per mole kelvin difference) molar free energy of formation Δ_fG° | aqueous | -628 kJ/mol (kilojoules per mole) molar heat of formation Δ_fH° | aqueous | -665 kJ/mol (kilojoules per mole) molar entropy S° | aqueous | -264 J/(mol K) (joules per mole kelvin)