Input interpretation
barium carbonate | lanthanum sulfide
Chemical names and formulas
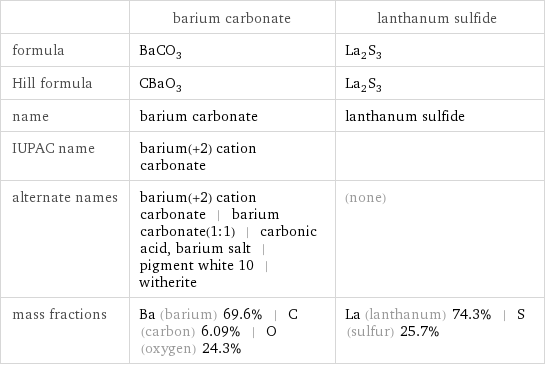
| barium carbonate | lanthanum sulfide formula | BaCO_3 | La_2S_3 Hill formula | CBaO_3 | La_2S_3 name | barium carbonate | lanthanum sulfide IUPAC name | barium(+2) cation carbonate | alternate names | barium(+2) cation carbonate | barium carbonate(1:1) | carbonic acid, barium salt | pigment white 10 | witherite | (none) mass fractions | Ba (barium) 69.6% | C (carbon) 6.09% | O (oxygen) 24.3% | La (lanthanum) 74.3% | S (sulfur) 25.7%
Structure diagrams

Structure diagrams
Basic properties
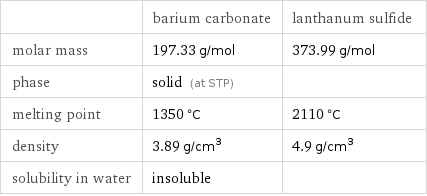
| barium carbonate | lanthanum sulfide molar mass | 197.33 g/mol | 373.99 g/mol phase | solid (at STP) | melting point | 1350 °C | 2110 °C density | 3.89 g/cm^3 | 4.9 g/cm^3 solubility in water | insoluble |
Units
