Input interpretation

rare earth metals | covalent radius
Summary
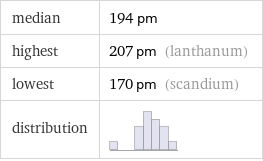
median | 194 pm highest | 207 pm (lanthanum) lowest | 170 pm (scandium) distribution |
Units

Plot
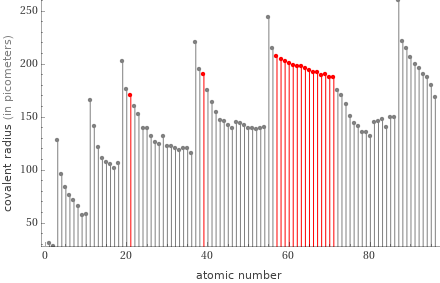
Plot
Periodic table location
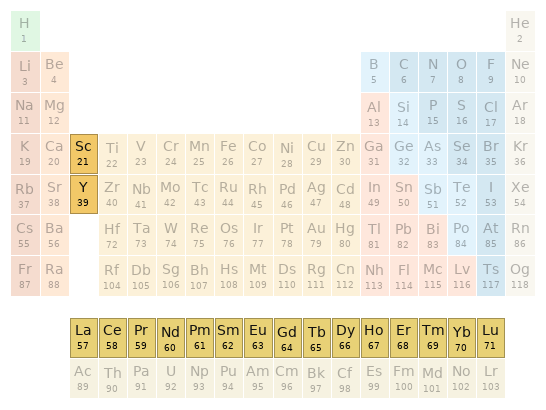
Periodic table location
Atomic radii properties

atomic radius (empirical) | median | 180 pm | highest | 195 pm (lanthanum) | lowest | 160 pm (scandium) | distribution | atomic radius (calculated) | median | 225 pm | highest | 247 pm (praseodymium) | lowest | 184 pm (scandium) | distribution | covalent radius | median | 194 pm | highest | 207 pm (lanthanum) | lowest | 170 pm (scandium) | distribution |
Units

Distribution plots
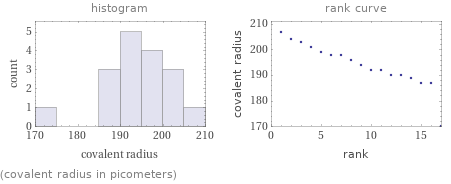
(covalent radius in picometers)
Covalent radius rankings
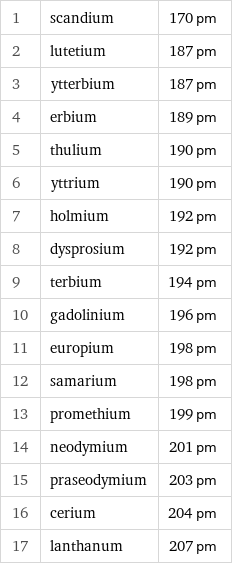
1 | scandium | 170 pm 2 | lutetium | 187 pm 3 | ytterbium | 187 pm 4 | erbium | 189 pm 5 | thulium | 190 pm 6 | yttrium | 190 pm 7 | holmium | 192 pm 8 | dysprosium | 192 pm 9 | terbium | 194 pm 10 | gadolinium | 196 pm 11 | europium | 198 pm 12 | samarium | 198 pm 13 | promethium | 199 pm 14 | neodymium | 201 pm 15 | praseodymium | 203 pm 16 | cerium | 204 pm 17 | lanthanum | 207 pm
Unit conversions for median covalent radius 194 pm
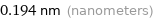
0.194 nm (nanometers)
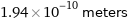
1.94×10^-10 meters
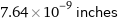
7.64×10^-9 inches
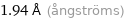
1.94 Å (ångströms)
Comparisons for median covalent radius 194 pm
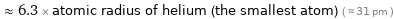
≈ 6.3 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
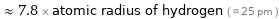
≈ 7.8 × atomic radius of hydrogen ( ≈ 25 pm )