Input interpretation

period 1 elements | molar volume
Results

hydrogen | 11200 cm^3/mol (cubic centimeters per mole) helium | 22420 cm^3/mol (cubic centimeters per mole)
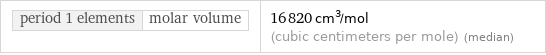
period 1 elements | molar volume | 16820 cm^3/mol (cubic centimeters per mole) (median)
Relative values

| visual | ratios | | comparisons hydrogen | | 0.5 | 1 | 50% smaller helium | | 1 | 2 | 100% larger
Unit conversions for median molar volume 16820 cm^3/mol
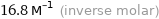
16.8 M^(-1) (inverse molar)
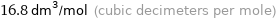
16.8 dm^3/mol (cubic decimeters per mole)
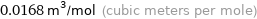
0.0168 m^3/mol (cubic meters per mole)

16.8 L/mol (liters per mole)
Comparison for median molar volume 16820 cm^3/mol
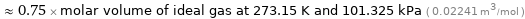
≈ 0.75 × molar volume of ideal gas at 273.15 K and 101.325 kPa ( 0.02241 m^3/mol )