Input interpretation

sodium thiocyanate | elemental composition
Result
Find the elemental composition for sodium thiocyanate in terms of the atom and mass percents: atom percent = N_i/N_atoms × 100% mass percent = (N_im_i)/m × 100% Plan: • Write the chemical formula and gather atomic masses from the periodic table. • Determine values for N_i, m_i, N_atoms and m using these items. • Finally, compute the percents and check the results. Write the chemical formula: NaSCN Use the chemical formula, NaSCN, to count the number of atoms, N_i, for each element and find the total number of atoms, N_atoms: | number of atoms C (carbon) | 1 N (nitrogen) | 1 Na (sodium) | 1 S (sulfur) | 1 N_atoms = 1 + 1 + 1 + 1 = 4 Divide each N_i by N_atoms to calculate atom fractions. Then use the property that atom fractions must sum to one to check the work: | number of atoms | atom fraction C (carbon) | 1 | 1/4 N (nitrogen) | 1 | 1/4 Na (sodium) | 1 | 1/4 S (sulfur) | 1 | 1/4 Check: 1/4 + 1/4 + 1/4 + 1/4 = 1 Compute atom percents using the atom fractions: | number of atoms | atom percent C (carbon) | 1 | 1/4 × 100% = 25.0% N (nitrogen) | 1 | 1/4 × 100% = 25.0% Na (sodium) | 1 | 1/4 × 100% = 25.0% S (sulfur) | 1 | 1/4 × 100% = 25.0% Look up the atomic mass, m_i, in unified atomic mass units, u, for each element in the periodic table: | number of atoms | atom percent | atomic mass/u C (carbon) | 1 | 25.0% | 12.011 N (nitrogen) | 1 | 25.0% | 14.007 Na (sodium) | 1 | 25.0% | 22.98976928 S (sulfur) | 1 | 25.0% | 32.06 Multiply N_i by m_i to compute the mass for each element. Then sum those values to compute the molecular mass, m: | number of atoms | atom percent | atomic mass/u | mass/u C (carbon) | 1 | 25.0% | 12.011 | 1 × 12.011 = 12.011 N (nitrogen) | 1 | 25.0% | 14.007 | 1 × 14.007 = 14.007 Na (sodium) | 1 | 25.0% | 22.98976928 | 1 × 22.98976928 = 22.98976928 S (sulfur) | 1 | 25.0% | 32.06 | 1 × 32.06 = 32.06 m = 12.011 u + 14.007 u + 22.98976928 u + 32.06 u = 81.06776928 u Divide the mass for each element by m to calculate mass fractions. Then use the property that mass fractions must sum to one to check the work: | number of atoms | atom percent | mass fraction C (carbon) | 1 | 25.0% | 12.011/81.06776928 N (nitrogen) | 1 | 25.0% | 14.007/81.06776928 Na (sodium) | 1 | 25.0% | 22.98976928/81.06776928 S (sulfur) | 1 | 25.0% | 32.06/81.06776928 Check: 12.011/81.06776928 + 14.007/81.06776928 + 22.98976928/81.06776928 + 32.06/81.06776928 = 1 Compute mass percents using the mass fractions: Answer: | | | number of atoms | atom percent | mass percent C (carbon) | 1 | 25.0% | 12.011/81.06776928 × 100% = 14.82% N (nitrogen) | 1 | 25.0% | 14.007/81.06776928 × 100% = 17.28% Na (sodium) | 1 | 25.0% | 22.98976928/81.06776928 × 100% = 28.36% S (sulfur) | 1 | 25.0% | 32.06/81.06776928 × 100% = 39.55%
Mass fraction pie chart
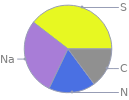
Mass fraction pie chart