Input interpretation
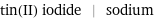
tin(II) iodide | sodium
Chemical names and formulas
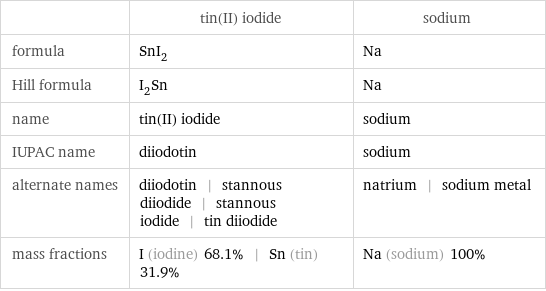
| tin(II) iodide | sodium formula | SnI_2 | Na Hill formula | I_2Sn | Na name | tin(II) iodide | sodium IUPAC name | diiodotin | sodium alternate names | diiodotin | stannous diiodide | stannous iodide | tin diiodide | natrium | sodium metal mass fractions | I (iodine) 68.1% | Sn (tin) 31.9% | Na (sodium) 100%
Structure diagrams
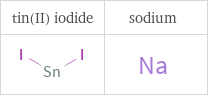
Structure diagrams
Basic properties
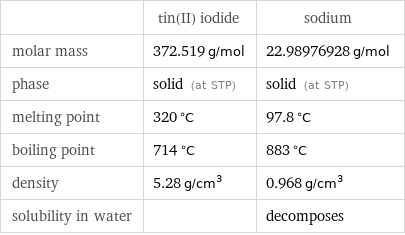
| tin(II) iodide | sodium molar mass | 372.519 g/mol | 22.98976928 g/mol phase | solid (at STP) | solid (at STP) melting point | 320 °C | 97.8 °C boiling point | 714 °C | 883 °C density | 5.28 g/cm^3 | 0.968 g/cm^3 solubility in water | | decomposes
Units
