Input interpretation
iodine monochloride | magnesium oxide
Chemical names and formulas
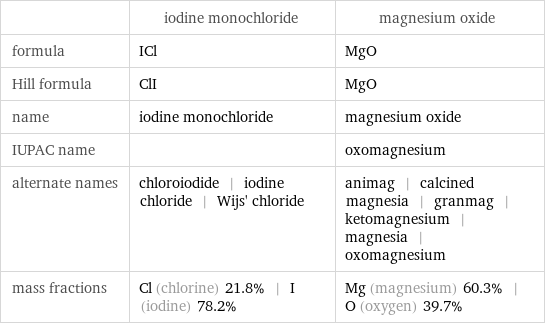
| iodine monochloride | magnesium oxide formula | ICl | MgO Hill formula | ClI | MgO name | iodine monochloride | magnesium oxide IUPAC name | | oxomagnesium alternate names | chloroiodide | iodine chloride | Wijs' chloride | animag | calcined magnesia | granmag | ketomagnesium | magnesia | oxomagnesium mass fractions | Cl (chlorine) 21.8% | I (iodine) 78.2% | Mg (magnesium) 60.3% | O (oxygen) 39.7%
Structure diagrams
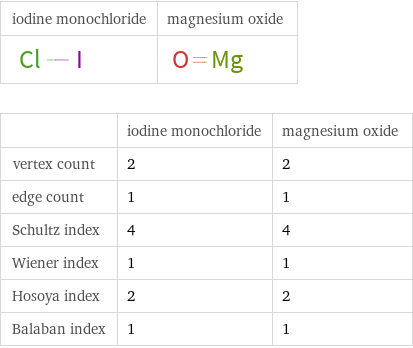
| iodine monochloride | magnesium oxide vertex count | 2 | 2 edge count | 1 | 1 Schultz index | 4 | 4 Wiener index | 1 | 1 Hosoya index | 2 | 2 Balaban index | 1 | 1
Basic properties
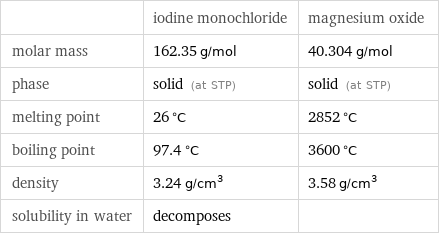
| iodine monochloride | magnesium oxide molar mass | 162.35 g/mol | 40.304 g/mol phase | solid (at STP) | solid (at STP) melting point | 26 °C | 2852 °C boiling point | 97.4 °C | 3600 °C density | 3.24 g/cm^3 | 3.58 g/cm^3 solubility in water | decomposes |
Units

Thermodynamic properties
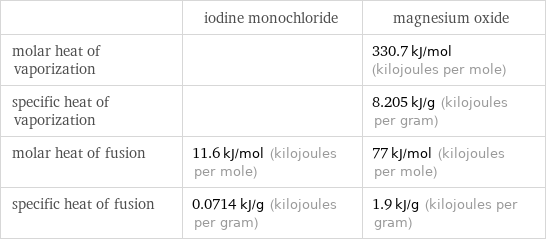
| iodine monochloride | magnesium oxide molar heat of vaporization | | 330.7 kJ/mol (kilojoules per mole) specific heat of vaporization | | 8.205 kJ/g (kilojoules per gram) molar heat of fusion | 11.6 kJ/mol (kilojoules per mole) | 77 kJ/mol (kilojoules per mole) specific heat of fusion | 0.0714 kJ/g (kilojoules per gram) | 1.9 kJ/g (kilojoules per gram)
Solid properties (at STP)

| iodine monochloride | magnesium oxide density | 3.24 g/cm^3 | 3.58 g/cm^3 vapor pressure | 30 mmHg | 0.3 mmHg
Units
