Input interpretation
cuprous chloride
Chemical names and formulas

formula | CuCl Hill formula | ClCu name | cuprous chloride alternate names | chlorocopper | copper chloride | copper(I) chloride | copper monochloride mass fractions | Cl (chlorine) 35.8% | Cu (copper) 64.2%
Structure diagram
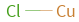
Structure diagram
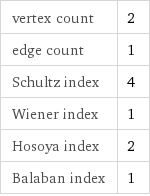
vertex count | 2 edge count | 1 Schultz index | 4 Wiener index | 1 Hosoya index | 2 Balaban index | 1
Basic properties
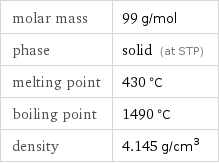
molar mass | 99 g/mol phase | solid (at STP) melting point | 430 °C boiling point | 1490 °C density | 4.145 g/cm^3
Units

Solid properties (at STP)
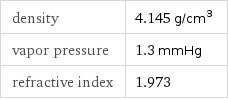
density | 4.145 g/cm^3 vapor pressure | 1.3 mmHg refractive index | 1.973
Units

Thermodynamic properties
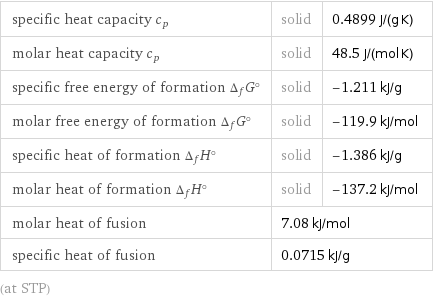
specific heat capacity c_p | solid | 0.4899 J/(g K) molar heat capacity c_p | solid | 48.5 J/(mol K) specific free energy of formation Δ_fG° | solid | -1.211 kJ/g molar free energy of formation Δ_fG° | solid | -119.9 kJ/mol specific heat of formation Δ_fH° | solid | -1.386 kJ/g molar heat of formation Δ_fH° | solid | -137.2 kJ/mol molar heat of fusion | 7.08 kJ/mol | specific heat of fusion | 0.0715 kJ/g | (at STP)
Chemical identifiers

CAS number | 7758-89-6 RTECS number | GL6990000 MDL number | MFCD00010971
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties
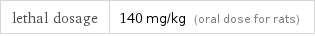
lethal dosage | 140 mg/kg (oral dose for rats)

probable lethal dose for man | 30 mL (milliliters) RTECS classes | agricultural chemical and pesticide | mutagen