Input interpretation
strontium hydroxide
Chemical names and formulas

formula | Sr(OH)_2 Hill formula | H_2O_2Sr name | strontium hydroxide IUPAC name | strontium dihydroxide alternate names | strontium dihydroxide mass fractions | H (hydrogen) 1.66% | O (oxygen) 26.3% | Sr (strontium) 72%
Structure diagram
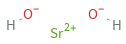
Structure diagram
Basic properties

molar mass | 121.6 g/mol
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -7.884 kJ/g molar heat of formation Δ_fH° | solid | -959 kJ/mol molar heat of fusion | 23 kJ/mol | specific heat of fusion | 0.19 kJ/g | (at STP)
Chemical identifiers
![CAS number | 18480-07-4 PubChem CID number | 87672 SMILES identifier | [OH-].[OH-].[Sr+2] InChI identifier | InChI=1/2H2O.Sr/h2*1H2;/q;;+2/p-2/f2HO.Sr/h2*1h;/q2*-1;m RTECS number | WK9100000 MDL number | MFCD00036273](../image_source/001ff367bf23c387307ea6ec84f432ae.png)
CAS number | 18480-07-4 PubChem CID number | 87672 SMILES identifier | [OH-].[OH-].[Sr+2] InChI identifier | InChI=1/2H2O.Sr/h2*1H2;/q;;+2/p-2/f2HO.Sr/h2*1h;/q2*-1;m RTECS number | WK9100000 MDL number | MFCD00036273
Ion equivalents

Sr^(2+) (strontium cation) | 1 (OH)^- (hydroxide anion) | 2