Input interpretation
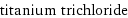
titanium trichloride
Chemical names and formulas

formula | TiCl_3 Hill formula | Cl_3Ti name | titanium trichloride IUPAC name | trichlorotitanium alternate names | titanium chloride | titanium(III) chloride | titanous chloride | trichlorotitanium mass fractions | Cl (chlorine) 69% | Ti (titanium) 31%
Structure diagram
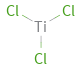
Structure diagram
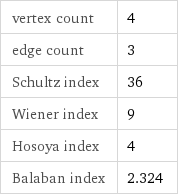
vertex count | 4 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties
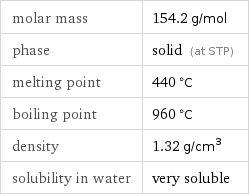
molar mass | 154.2 g/mol phase | solid (at STP) melting point | 440 °C boiling point | 960 °C density | 1.32 g/cm^3 solubility in water | very soluble
Units

Solid properties (at STP)
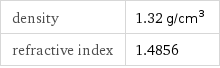
density | 1.32 g/cm^3 refractive index | 1.4856
Units

Thermodynamic properties
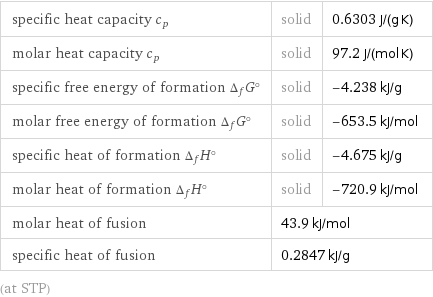
specific heat capacity c_p | solid | 0.6303 J/(g K) molar heat capacity c_p | solid | 97.2 J/(mol K) specific free energy of formation Δ_fG° | solid | -4.238 kJ/g molar free energy of formation Δ_fG° | solid | -653.5 kJ/mol specific heat of formation Δ_fH° | solid | -4.675 kJ/g molar heat of formation Δ_fH° | solid | -720.9 kJ/mol molar heat of fusion | 43.9 kJ/mol | specific heat of fusion | 0.2847 kJ/g | (at STP)
Chemical identifiers
Cl InChI identifier | InChI=1/3ClH.Ti/h3*1H;/q;;;+3/p-3/f3Cl.Ti/h3*1h;/q3*-1;m RTECS number | XR1924000 MDL number | MFCD00011266](../image_source/d9fee7a51d177f9c729f67d658d9e3c1.png)
CAS number | 7705-07-9 PubChem CID number | 62646 PubChem SID number | 24853202 SMILES identifier | Cl[Ti](Cl)Cl InChI identifier | InChI=1/3ClH.Ti/h3*1H;/q;;;+3/p-3/f3Cl.Ti/h3*1h;/q3*-1;m RTECS number | XR1924000 MDL number | MFCD00011266
Toxicity properties

RTECS classes | reproductive effector