Input interpretation
methyl tert-butyl ether
Chemical names and formulas

formula | (CH_3)_3COCH_3 Hill formula | C_5H_12O name | methyl tert-butyl ether IUPAC name | 2-methoxy-2-methylpropane alternate names | 2-methoxy-2-methyl-propane | 2-methoxy-2-methylpropane | ether, tert-butyl methyl | MTBE | propane, 2-methoxy-2-methyl- | tert-butyl methyl ether mass fractions | C (carbon) 68.1% | H (hydrogen) 13.7% | O (oxygen) 18.1%
Lewis structure
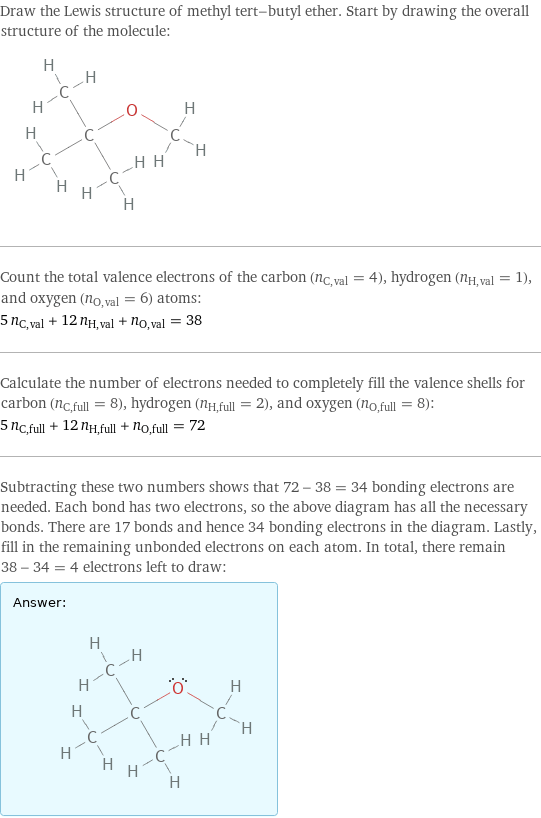
Draw the Lewis structure of methyl tert-butyl ether. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 5 n_C, val + 12 n_H, val + n_O, val = 38 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 5 n_C, full + 12 n_H, full + n_O, full = 72 Subtracting these two numbers shows that 72 - 38 = 34 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 17 bonds and hence 34 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 38 - 34 = 4 electrons left to draw: Answer: | |
3D structure
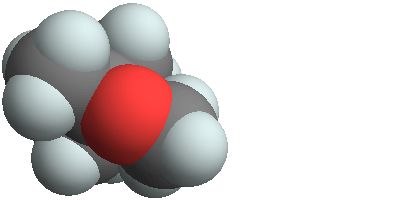
3D structure
Basic properties
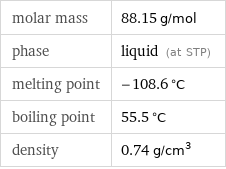
molar mass | 88.15 g/mol phase | liquid (at STP) melting point | -108.6 °C boiling point | 55.5 °C density | 0.74 g/cm^3
Units

Liquid properties (at STP)
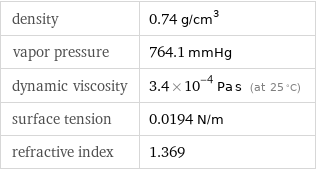
density | 0.74 g/cm^3 vapor pressure | 764.1 mmHg dynamic viscosity | 3.4×10^-4 Pa s (at 25 °C) surface tension | 0.0194 N/m refractive index | 1.369
Units
Thermodynamic properties
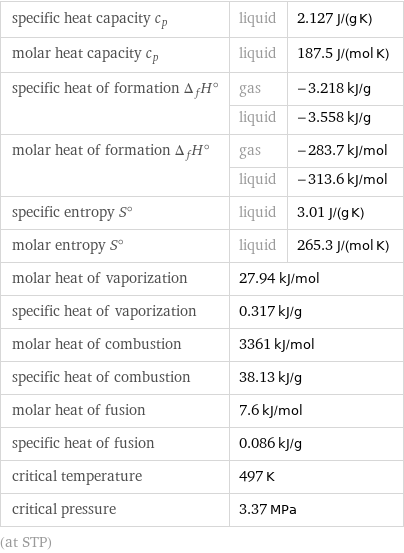
specific heat capacity c_p | liquid | 2.127 J/(g K) molar heat capacity c_p | liquid | 187.5 J/(mol K) specific heat of formation Δ_fH° | gas | -3.218 kJ/g | liquid | -3.558 kJ/g molar heat of formation Δ_fH° | gas | -283.7 kJ/mol | liquid | -313.6 kJ/mol specific entropy S° | liquid | 3.01 J/(g K) molar entropy S° | liquid | 265.3 J/(mol K) molar heat of vaporization | 27.94 kJ/mol | specific heat of vaporization | 0.317 kJ/g | molar heat of combustion | 3361 kJ/mol | specific heat of combustion | 38.13 kJ/g | molar heat of fusion | 7.6 kJ/mol | specific heat of fusion | 0.086 kJ/g | critical temperature | 497 K | critical pressure | 3.37 MPa | (at STP)
Chemical identifiers

CAS number | 1634-04-4 Beilstein number | 1730942 PubChem CID number | 15413 PubChem SID number | 24850654 SMILES identifier | CC(C)(C)OC InChI identifier | InChI=1/C5H12O/c1-5(2, 3)6-4/h1-4H3 RTECS number | KN5250000 MDL number | MFCD00008812
NFPA label

NFPA label

NFPA health rating | 2 NFPA fire rating | 3 NFPA reactivity rating | 0
Safety properties
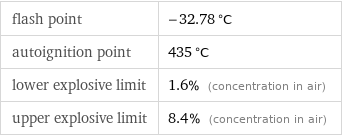
flash point | -32.78 °C autoignition point | 435 °C lower explosive limit | 1.6% (concentration in air) upper explosive limit | 8.4% (concentration in air)

DOT hazard class | 3 DOT numbers | 2398
Toxicity properties

threshold limit value | 50 ppmv

RTECS classes | tumorigen | mutagen | reproductive effector