Input interpretation

tellurium tetrabromide
Chemical names and formulas

formula | TeBr_4 Hill formula | Br_4Te name | tellurium tetrabromide mass fractions | Br (bromine) 71.5% | Te (tellurium) 28.5%
Lewis structure

Draw the Lewis structure of tellurium tetrabromide. Start by drawing the overall structure of the molecule: Count the total valence electrons of the bromine (n_Br, val = 7) and tellurium (n_Te, val = 6) atoms: 4 n_Br, val + n_Te, val = 34 Calculate the number of electrons needed to completely fill the valence shells for bromine (n_Br, full = 8) and tellurium (n_Te, full = 8): 4 n_Br, full + n_Te, full = 40 Subtracting these two numbers shows that 40 - 34 = 6 bonding electrons are needed, which are already accounted for in the structure. Note that the valence shell of tellurium has been expanded. After accounting for the expanded valence, there are 4 bonds and hence 8 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 34 - 8 = 26 electrons left to draw: Answer: | |
Basic properties
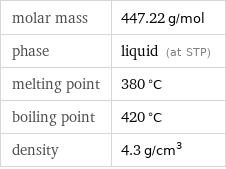
molar mass | 447.22 g/mol phase | liquid (at STP) melting point | 380 °C boiling point | 420 °C density | 4.3 g/cm^3
Units

Liquid properties (at STP)

density | 4.3 g/cm^3
Units

Thermodynamic properties
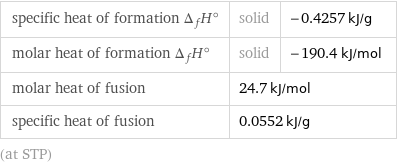
specific heat of formation Δ_fH° | solid | -0.4257 kJ/g molar heat of formation Δ_fH° | solid | -190.4 kJ/mol molar heat of fusion | 24.7 kJ/mol | specific heat of fusion | 0.0552 kJ/g | (at STP)
Chemical identifiers
(Br)Br InChI identifier | InChI=1/Br4Te/c1-5(2, 3)4 MDL number | MFCD00049575](../image_source/ff0bfcdd4ccffbef918963bf31ca1ca0.png)
CAS number | 10031-27-3 PubChem CID number | 82311 SMILES identifier | Br[Te](Br)(Br)Br InChI identifier | InChI=1/Br4Te/c1-5(2, 3)4 MDL number | MFCD00049575