Input interpretation
rubidium fluoride
Chemical names and formulas

formula | RbF Hill formula | FRb name | rubidium fluoride IUPAC name | rubidium(+1) cation fluoride alternate names | rubidium(+1) cation fluoride | rubidium monofluoride mass fractions | F (fluorine) 18.2% | Rb (rubidium) 81.8%
Structure diagram

Structure diagram
Basic properties
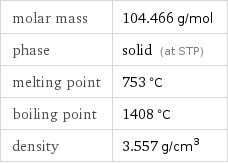
molar mass | 104.466 g/mol phase | solid (at STP) melting point | 753 °C boiling point | 1408 °C density | 3.557 g/cm^3
Units

Solid properties (at STP)

density | 3.557 g/cm^3 vapor pressure | 98.9 mmHg (at 1166 °C)
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -5.339 kJ/g specific heat of fusion | 0.247 kJ/g | (at STP)
Units

Chemical identifiers
![CAS number | 13446-74-7 PubChem CID number | 83473 PubChem SID number | 24855132 SMILES identifier | [F-].[Rb+]](../image_source/a50f207e64fc97fdb080052f6a24c74b.png)
CAS number | 13446-74-7 PubChem CID number | 83473 PubChem SID number | 24855132 SMILES identifier | [F-].[Rb+]