Input interpretation
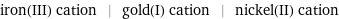
iron(III) cation | gold(I) cation | nickel(II) cation
Structure diagrams

Structure diagrams
General properties

| iron(III) cation | gold(I) cation | nickel(II) cation formula | Fe^(3+) | Au^+ | Ni^(2+) net ionic charge | +3 | +1 | +2 alternate names | ferric | iron(III) | iron(3+) | gold(I) | gold(1+) | nickelous | nickel(II) | nickel(2+)
Ionic radii
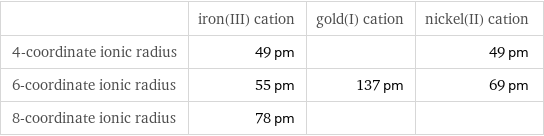
| iron(III) cation | gold(I) cation | nickel(II) cation 4-coordinate ionic radius | 49 pm | | 49 pm 6-coordinate ionic radius | 55 pm | 137 pm | 69 pm 8-coordinate ionic radius | 78 pm | |
Units

Other properties

| iron(III) cation | gold(I) cation | nickel(II) cation ion class | biomolecule ions | cations | d block ions | group 8 ions | iron(III) ions | monatomic cations | transition metal ions | cations | d block ions | gold(I) ions | group 11 ions | transition metal ions | biomolecule ions | cations | d block ions | group 10 ions | nickel(II) ions | transition metal ions common sources of ion | iron phosphide (3:1) (1 eq) | iron(III) sulfate hydrate (2 eq) | iron(III) phosphate tetrahydrate (1 eq) | iron(III) perchlorate hydrate (1 eq) | iron(III) oxalate hexahydrate (2 eq) | iron(III) nitrate nonahydrate (1 eq) | hemin (1 eq) | diironnonacarbonyl (1 eq) | gold(I) sulfide (1 eq) | gold cyanide (1 eq) | gold(I) chloride (1 eq) | nickel(II) sulfate hexahydrate (1 eq) | nickel(II) sulfate heptahydrate (1 eq) | nickel(II) sulfamate tetrahydrate (1 eq) | nickel(II) stearate (1 eq) | nickel(II) phthalocyanine (1 eq) | nickel(II) oxalate dihydrate (1 eq) | nickel(II) nitrate hexahydrate (1 eq) | nickel(II) molybdate (1 eq) | nickel(II) bis(4-cyclohexylbutyrate) (1 eq) | nickel chromium oxide (1 eq)