Input interpretation

potassium chloride | element types
Result
chlorine | potassium
Periodic table location
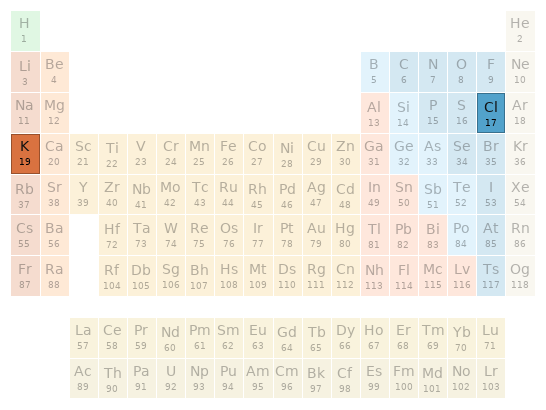
Periodic table location
Images

Images
Basic elemental properties
![| chlorine | potassium atomic symbol | Cl | K atomic number | 17 | 19 short electronic configuration | [Ne]3s^23p^5 | [Ar]4s^1 Aufbau diagram | 3p 3s | 4s block | p | s group | 17 | 1 period | 3 | 4 atomic mass | 35.45 u | 39.0983 u half-life | (stable) | (stable)](../image_source/af5cdc3cb03592ede9690c294d111b2a.png)
| chlorine | potassium atomic symbol | Cl | K atomic number | 17 | 19 short electronic configuration | [Ne]3s^23p^5 | [Ar]4s^1 Aufbau diagram | 3p 3s | 4s block | p | s group | 17 | 1 period | 3 | 4 atomic mass | 35.45 u | 39.0983 u half-life | (stable) | (stable)
Thermodynamic properties
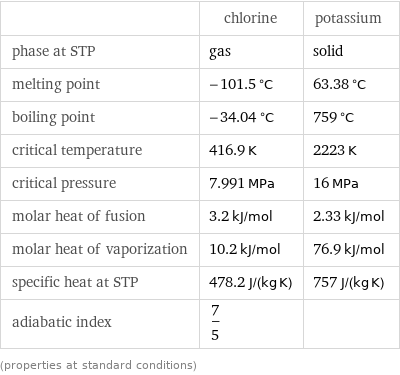
| chlorine | potassium phase at STP | gas | solid melting point | -101.5 °C | 63.38 °C boiling point | -34.04 °C | 759 °C critical temperature | 416.9 K | 2223 K critical pressure | 7.991 MPa | 16 MPa molar heat of fusion | 3.2 kJ/mol | 2.33 kJ/mol molar heat of vaporization | 10.2 kJ/mol | 76.9 kJ/mol specific heat at STP | 478.2 J/(kg K) | 757 J/(kg K) adiabatic index | 7/5 | (properties at standard conditions)
Material properties
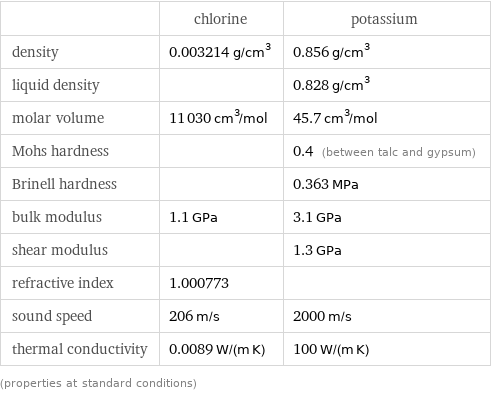
| chlorine | potassium density | 0.003214 g/cm^3 | 0.856 g/cm^3 liquid density | | 0.828 g/cm^3 molar volume | 11030 cm^3/mol | 45.7 cm^3/mol Mohs hardness | | 0.4 (between talc and gypsum) Brinell hardness | | 0.363 MPa bulk modulus | 1.1 GPa | 3.1 GPa shear modulus | | 1.3 GPa refractive index | 1.000773 | sound speed | 206 m/s | 2000 m/s thermal conductivity | 0.0089 W/(m K) | 100 W/(m K) (properties at standard conditions)
Electromagnetic properties
| chlorine | potassium electrical type | insulator | conductor resistivity | 100 Ω m (ohm meters) | 7×10^-8 Ω m (ohm meters) electrical conductivity | 0.01 S/m (siemens per meter) | 1.4×10^7 S/m (siemens per meter) magnetic type | diamagnetic | paramagnetic volume magnetic susceptibility | -2.31×10^-8 | 5.74×10^-6 mass magnetic susceptibility | -7.2×10^-9 m^3/kg (cubic meters per kilogram) | 6.7×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -5.11×10^-10 m^3/mol (cubic meters per mole) | 2.62×10^-10 m^3/mol (cubic meters per mole) work function | | 2.29 eV (Polycrystalline) threshold frequency | | 5.537×10^14 Hz (hertz) color | (yellow) | (silver) refractive index | 1.000773 |
Reactivity
| chlorine | potassium valence | 5 | 1 electronegativity | 3.16 | 0.82 electron affinity | 349 kJ/mol (kilojoules per mole) | 48.4 kJ/mol (kilojoules per mole) first ionization energy | 1251.2 kJ/mol (kilojoules per mole) | 418.8 kJ/mol (kilojoules per mole) ionization energies | 1251.2 kJ/mol | 2298 kJ/mol | 3822 kJ/mol | 5158.6 kJ/mol | 6542 kJ/mol | 9362 kJ/mol | 11018 kJ/mol | 33604 kJ/mol | 38600 kJ/mol | 43961 kJ/mol | 418.8 kJ/mol | 3052 kJ/mol | 4420 kJ/mol | 5877 kJ/mol | 7975 kJ/mol | 9590 kJ/mol | 11343 kJ/mol | 14944 kJ/mol | 16963.7 kJ/mol | 48610 kJ/mol
Reactivity
Atomic properties
| chlorine | potassium term symbol | ^2P_(3/2) | ^2S_(1/2) atomic radius | 100 pm | 220 pm covalent radius | 102 pm | 203 pm van der Waals radius | 175 pm | 275 pm (electronic ground state properties)
Abundances
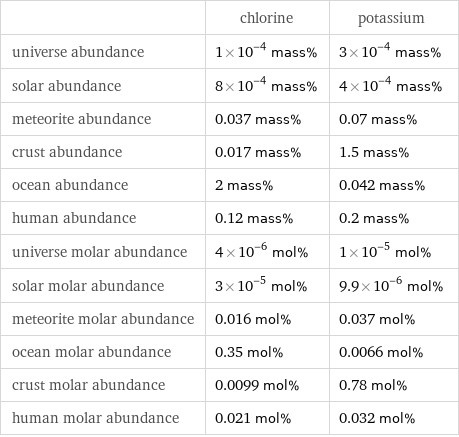
| chlorine | potassium universe abundance | 1×10^-4 mass% | 3×10^-4 mass% solar abundance | 8×10^-4 mass% | 4×10^-4 mass% meteorite abundance | 0.037 mass% | 0.07 mass% crust abundance | 0.017 mass% | 1.5 mass% ocean abundance | 2 mass% | 0.042 mass% human abundance | 0.12 mass% | 0.2 mass% universe molar abundance | 4×10^-6 mol% | 1×10^-5 mol% solar molar abundance | 3×10^-5 mol% | 9.9×10^-6 mol% meteorite molar abundance | 0.016 mol% | 0.037 mol% ocean molar abundance | 0.35 mol% | 0.0066 mol% crust molar abundance | 0.0099 mol% | 0.78 mol% human molar abundance | 0.021 mol% | 0.032 mol%
Nuclear properties

| chlorine | potassium half-life | (stable) | (stable) stable isotopes | Cl-35 (75.77%) | Cl-37 (24.23%) | K-39 (93.2581%) | K-41 (6.7302%) nuclear spin | Cl-28: 1^+ | Cl-29: 3/2^+ | Cl-30: 3^+ | Cl-31: 3/2^+ | Cl-32: 1^+ | Cl-33: 3/2^+ | Cl-34: 0^+ | Cl-35: 3/2^+ | Cl-36: 2^+ | Cl-37: 3/2^+ | Cl-38: 2^- | Cl-39: 3/2^+ | Cl-40: 2^- | Cl-41: 1/2^+ | Cl-43: 1/2^+ | Cl-45: 1/2^+ | Cl-47: 3/2^+ | Cl-49: 3/2^+ | Cl-51: 3/2^+ | K-32: 1^+ | K-33: 3/2^+ | K-34: 1^+ | K-35: 3/2^+ | K-36: 2^+ | K-37: 3/2^+ | K-38: 3^+ | K-39: 3/2^+ | K-40: 4^- | K-41: 3/2^+ | K-42: 2^- | K-43: 3/2^+ | K-44: 2^- | K-45: 3/2^+ | K-46: 2^- | K-47: 1/2^+ | K-48: 2^- | K-49: 3/2^+ | K-52: 2^- | K-53: 3/2^+ | K-54: 2^- | K-55: 3/2^+ unstable isotopes | Cl-36 (0.3 Myr) | Cl-39 (56.2 min) | Cl-38 (37.23 min) | Cl-40 (81 s) | Cl-41 (38.4 s) | Cl-42 (6.8 s) | Cl-43 (3.13 s) | Cl-33 (2.511 s) | Cl-34 (1.5264 s) | Cl-44 (560 ms) | Cl-45 (413 ms) | Cl-32 (298 ms) | Cl-46 (232 ms) | Cl-31 (150 ms) | Cl-47 (101 ms) | Cl-50 (20 ms) | Cl-51 (200 ns) | Cl-48 (200 ns) | Cl-49 (170 ns) | Cl-30 (30 ns) | Cl-29 (20 ns) | K-40 (1.248 Gyr) | K-43 (22.3 h) | K-42 (12.321 h) | K-44 (22.13 min) | K-45 (17.81 min) | K-38 (7.637 min) | K-46 (105 s) | K-47 (17.5 s) | K-48 (6.8 s) | K-49 (1.26 s) | K-37 (1.226 s) | K-50 (472 ms) | K-51 (365 ms) | K-36 (342 ms) | K-35 (178 ms) | K-52 (105 ms) | K-53 (30 ms) | K-54 (10 ms) | K-55 (3 ms) | K-34 (25 ns) | K-33 (25 ns) neutron cross-section | 35.3 b | 2.1 b neutron mass absorption | 0.033 m^2/kg | 0.0018 m^2/kg
Identifiers
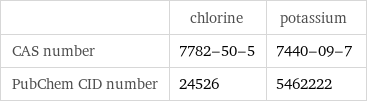
| chlorine | potassium CAS number | 7782-50-5 | 7440-09-7 PubChem CID number | 24526 | 5462222