Input interpretation
sodium peroxide
Chemical names and formulas

formula | Na_2O_2 name | sodium peroxide IUPAC name | disodium peroxide alternate names | disodium dioxide | disodium peroxide | sodium dioxide mass fractions | Na (sodium) 59% | O (oxygen) 41%
Structure diagram

Structure diagram
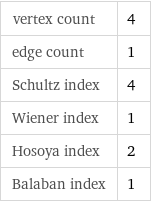
vertex count | 4 edge count | 1 Schultz index | 4 Wiener index | 1 Hosoya index | 2 Balaban index | 1
Basic properties
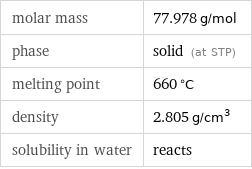
molar mass | 77.978 g/mol phase | solid (at STP) melting point | 660 °C density | 2.805 g/cm^3 solubility in water | reacts
Units

Solid properties (at STP)

density | 2.805 g/cm^3
Units

Thermodynamic properties

specific heat capacity c_p | solid | 1.144 J/(g K) specific free energy of formation Δ_fG° | solid | -5.741 kJ/g specific heat of formation Δ_fH° | solid | -6.552 kJ/g specific entropy S° | solid | 1.218 J/(g K) specific heat of fusion | 0.314 kJ/g | (at STP)
Units

Chemical identifiers
![CAS number | 1313-60-6 PubChem CID number | 14803 SMILES identifier | [O-][O-].[Na+].[Na+]](../image_source/db882e348937357e289e8f98999fd900.png)
CAS number | 1313-60-6 PubChem CID number | 14803 SMILES identifier | [O-][O-].[Na+].[Na+]
NFPA label

NFPA label
Ion equivalents
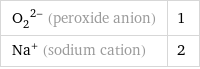
(O_2)^(2-) (peroxide anion) | 1 Na^+ (sodium cation) | 2