Input interpretation
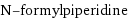
N-formylpiperidine
Chemical names and formulas

formula | C_6H_11NO name | N-formylpiperidine IUPAC name | 1-piperidinecarboxaldehyde alternate names | 1-formylpiperidine | 1-piperidinecarboxaldehyde | piperidine-1-carbaldehyde | piperidine-1-carboxaldehyde | piperidine-N-carbaldehyde mass fractions | C (carbon) 63.7% | H (hydrogen) 9.8% | N (nitrogen) 12.4% | O (oxygen) 14.1%
Lewis structure
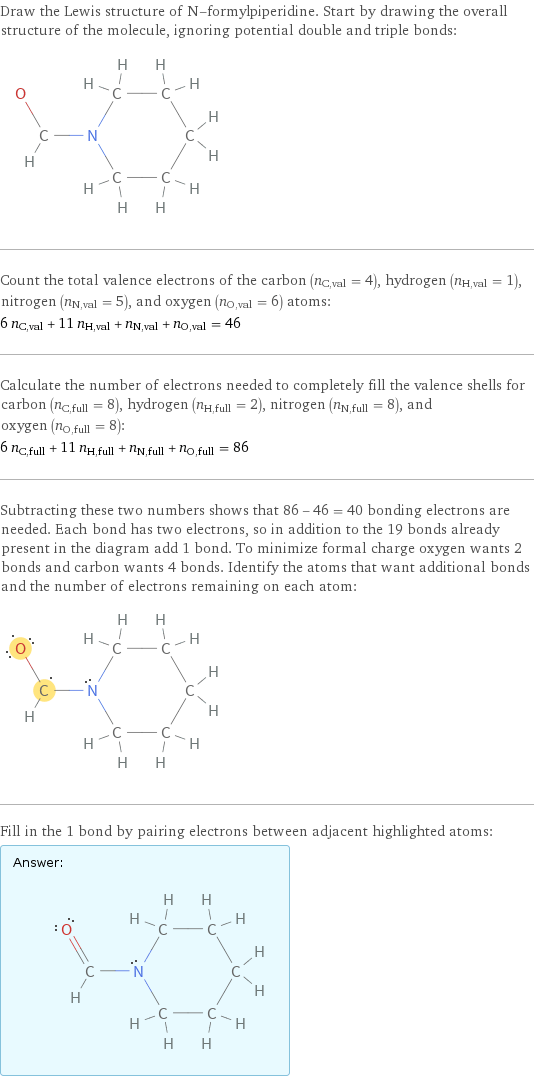
Draw the Lewis structure of N-formylpiperidine. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), nitrogen (n_N, val = 5), and oxygen (n_O, val = 6) atoms: 6 n_C, val + 11 n_H, val + n_N, val + n_O, val = 46 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), nitrogen (n_N, full = 8), and oxygen (n_O, full = 8): 6 n_C, full + 11 n_H, full + n_N, full + n_O, full = 86 Subtracting these two numbers shows that 86 - 46 = 40 bonding electrons are needed. Each bond has two electrons, so in addition to the 19 bonds already present in the diagram add 1 bond. To minimize formal charge oxygen wants 2 bonds and carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 1 bond by pairing electrons between adjacent highlighted atoms: Answer: | |
3D structure
3D structure
Basic properties
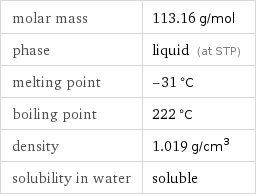
molar mass | 113.16 g/mol phase | liquid (at STP) melting point | -31 °C boiling point | 222 °C density | 1.019 g/cm^3 solubility in water | soluble
Units

Hydrophobicity and permeability properties

predicted LogP hydrophobicity | 0.21 predicted LogS | 0.4
Basic drug properties
approval status | experimental | small molecule
Liquid properties (at STP)
density | 1.019 g/cm^3 vapor pressure | 0.09998 mmHg refractive index | 1.484
Units

Chemical identifiers

CAS number | 2591-86-8 Beilstein number | 107697 PubChem CID number | 17429 PubChem SID number | 24894793 SMILES identifier | C1CCN(CC1)C=O InChI identifier | InChI=1/C6H11NO/c8-6-7-4-2-1-3-5-7/h6H, 1-5H2 InChI key | FEWLNYSYJNLUOO-UHFFFAOYAJ RTECS number | TN0380000 MDL number | MFCD00006483
NFPA label

NFPA label

NFPA health rating | 2 NFPA fire rating | 2 NFPA reactivity rating | 0
Safety properties

flash point | 91.67 °C
DOT hazard class | 3 DOT numbers | 1993
Toxicity properties

RTECS classes | reproductive effector | primary irritant