Input interpretation
mixed sulfur
Chemical names and formulas

formula | S name | mixed sulfur IUPAC name | sulfur alternate names | brimstone | elemental sulfur | mixed sulfur allotropes | precipitated sulfur mass fractions | S (sulfur) 100%
Structure diagram

Structure diagram
Basic properties
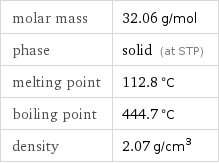
molar mass | 32.06 g/mol phase | solid (at STP) melting point | 112.8 °C boiling point | 444.7 °C density | 2.07 g/cm^3
Units

Solid properties (at STP)
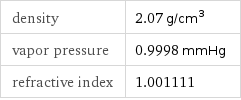
density | 2.07 g/cm^3 vapor pressure | 0.9998 mmHg refractive index | 1.001111
Units

Thermodynamic properties

specific heat of vaporization | 0.2776 kJ/g specific heat of fusion | 0.03743 kJ/g (at STP)
Units

Chemical identifiers
![CAS number | 7704-34-9 PubChem CID number | 5362487 PubChem SID number | 10535436 SMILES identifier | [S]](../image_source/404fd3c2f9b55459bbee8c424149e2d7.png)
CAS number | 7704-34-9 PubChem CID number | 5362487 PubChem SID number | 10535436 SMILES identifier | [S]