Input interpretation
carbon (chemical element) | hydrogen (chemical element) | thallium (chemical element)
Periodic table location
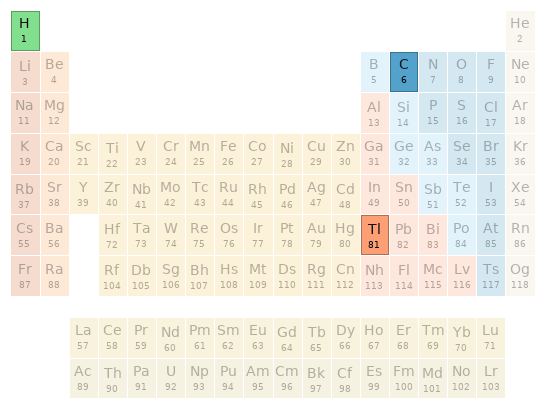
Periodic table location
Images

Images
Basic elemental properties
![| carbon | hydrogen | thallium atomic symbol | C | H | Tl atomic number | 6 | 1 | 81 short electronic configuration | [He]2s^22p^2 | 1s^1 | [Xe]6s^24f^145d^106p^1 Aufbau diagram | 2p 2s | 1s | 6p 5d 4f 6s block | p | s | p group | 14 | 1 | 13 period | 2 | 1 | 6 atomic mass | 12.011 u | 1.008 u | 204.38 u half-life | (stable) | (stable) | (stable)](../image_source/bca24b4a3d79e875c30dda89b42c5919.png)
| carbon | hydrogen | thallium atomic symbol | C | H | Tl atomic number | 6 | 1 | 81 short electronic configuration | [He]2s^22p^2 | 1s^1 | [Xe]6s^24f^145d^106p^1 Aufbau diagram | 2p 2s | 1s | 6p 5d 4f 6s block | p | s | p group | 14 | 1 | 13 period | 2 | 1 | 6 atomic mass | 12.011 u | 1.008 u | 204.38 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
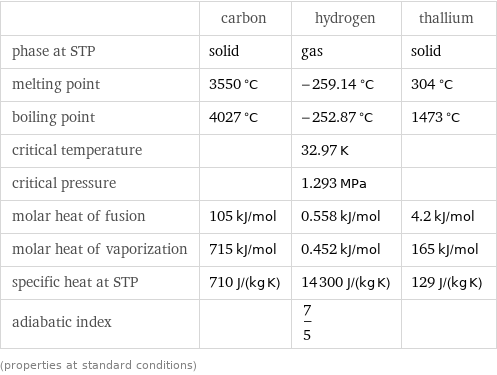
| carbon | hydrogen | thallium phase at STP | solid | gas | solid melting point | 3550 °C | -259.14 °C | 304 °C boiling point | 4027 °C | -252.87 °C | 1473 °C critical temperature | | 32.97 K | critical pressure | | 1.293 MPa | molar heat of fusion | 105 kJ/mol | 0.558 kJ/mol | 4.2 kJ/mol molar heat of vaporization | 715 kJ/mol | 0.452 kJ/mol | 165 kJ/mol specific heat at STP | 710 J/(kg K) | 14300 J/(kg K) | 129 J/(kg K) adiabatic index | | 7/5 | (properties at standard conditions)
Material properties
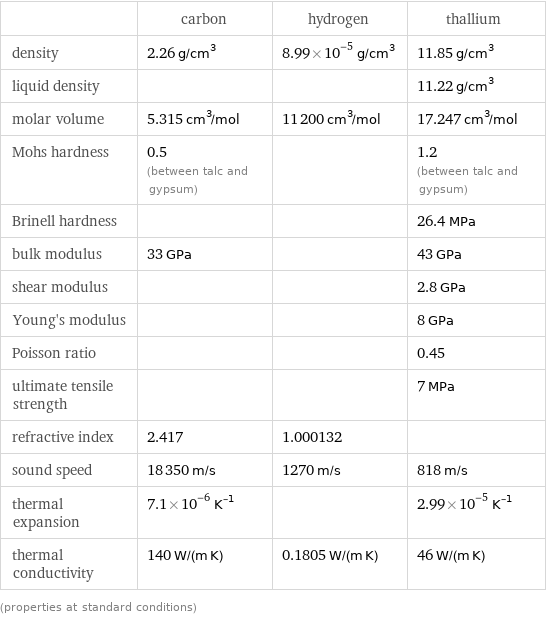
| carbon | hydrogen | thallium density | 2.26 g/cm^3 | 8.99×10^-5 g/cm^3 | 11.85 g/cm^3 liquid density | | | 11.22 g/cm^3 molar volume | 5.315 cm^3/mol | 11200 cm^3/mol | 17.247 cm^3/mol Mohs hardness | 0.5 (between talc and gypsum) | | 1.2 (between talc and gypsum) Brinell hardness | | | 26.4 MPa bulk modulus | 33 GPa | | 43 GPa shear modulus | | | 2.8 GPa Young's modulus | | | 8 GPa Poisson ratio | | | 0.45 ultimate tensile strength | | | 7 MPa refractive index | 2.417 | 1.000132 | sound speed | 18350 m/s | 1270 m/s | 818 m/s thermal expansion | 7.1×10^-6 K^(-1) | | 2.99×10^-5 K^(-1) thermal conductivity | 140 W/(m K) | 0.1805 W/(m K) | 46 W/(m K) (properties at standard conditions)
Electromagnetic properties
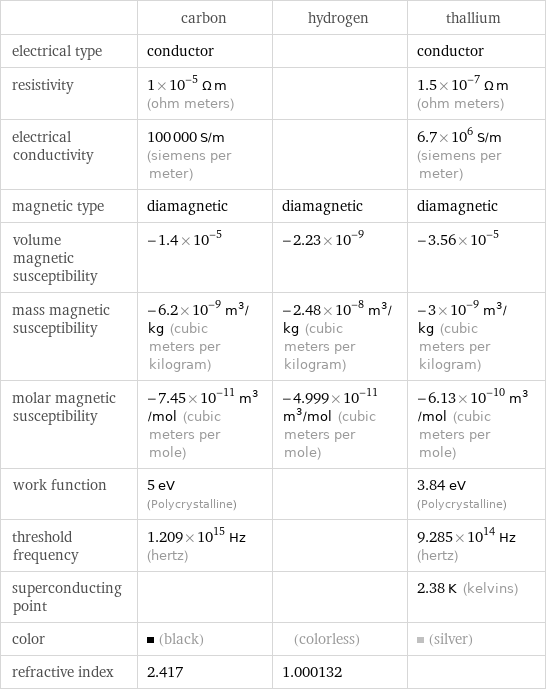
| carbon | hydrogen | thallium electrical type | conductor | | conductor resistivity | 1×10^-5 Ω m (ohm meters) | | 1.5×10^-7 Ω m (ohm meters) electrical conductivity | 100000 S/m (siemens per meter) | | 6.7×10^6 S/m (siemens per meter) magnetic type | diamagnetic | diamagnetic | diamagnetic volume magnetic susceptibility | -1.4×10^-5 | -2.23×10^-9 | -3.56×10^-5 mass magnetic susceptibility | -6.2×10^-9 m^3/kg (cubic meters per kilogram) | -2.48×10^-8 m^3/kg (cubic meters per kilogram) | -3×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -7.45×10^-11 m^3/mol (cubic meters per mole) | -4.999×10^-11 m^3/mol (cubic meters per mole) | -6.13×10^-10 m^3/mol (cubic meters per mole) work function | 5 eV (Polycrystalline) | | 3.84 eV (Polycrystalline) threshold frequency | 1.209×10^15 Hz (hertz) | | 9.285×10^14 Hz (hertz) superconducting point | | | 2.38 K (kelvins) color | (black) | (colorless) | (silver) refractive index | 2.417 | 1.000132 |
Reactivity
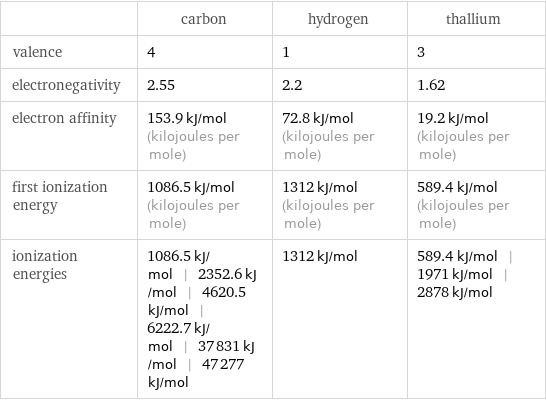
| carbon | hydrogen | thallium valence | 4 | 1 | 3 electronegativity | 2.55 | 2.2 | 1.62 electron affinity | 153.9 kJ/mol (kilojoules per mole) | 72.8 kJ/mol (kilojoules per mole) | 19.2 kJ/mol (kilojoules per mole) first ionization energy | 1086.5 kJ/mol (kilojoules per mole) | 1312 kJ/mol (kilojoules per mole) | 589.4 kJ/mol (kilojoules per mole) ionization energies | 1086.5 kJ/mol | 2352.6 kJ/mol | 4620.5 kJ/mol | 6222.7 kJ/mol | 37831 kJ/mol | 47277 kJ/mol | 1312 kJ/mol | 589.4 kJ/mol | 1971 kJ/mol | 2878 kJ/mol
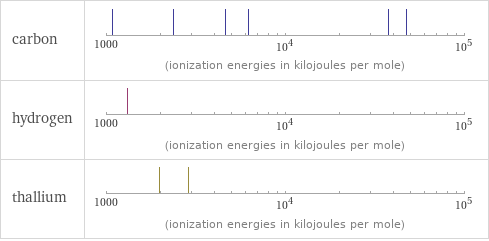
Reactivity
Atomic properties

| carbon | hydrogen | thallium term symbol | ^3P_0 | ^2S_(1/2) | ^2P_(1/2) atomic radius | 70 pm | 25 pm | 190 pm covalent radius | 76 pm | 31 pm | 145 pm van der Waals radius | 170 pm | 120 pm | 196 pm (electronic ground state properties)
Abundances
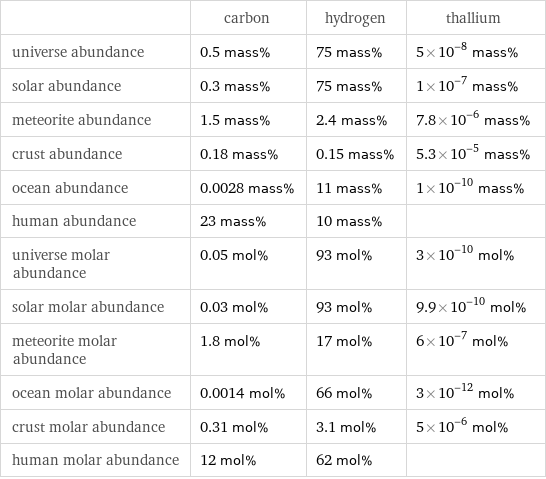
| carbon | hydrogen | thallium universe abundance | 0.5 mass% | 75 mass% | 5×10^-8 mass% solar abundance | 0.3 mass% | 75 mass% | 1×10^-7 mass% meteorite abundance | 1.5 mass% | 2.4 mass% | 7.8×10^-6 mass% crust abundance | 0.18 mass% | 0.15 mass% | 5.3×10^-5 mass% ocean abundance | 0.0028 mass% | 11 mass% | 1×10^-10 mass% human abundance | 23 mass% | 10 mass% | universe molar abundance | 0.05 mol% | 93 mol% | 3×10^-10 mol% solar molar abundance | 0.03 mol% | 93 mol% | 9.9×10^-10 mol% meteorite molar abundance | 1.8 mol% | 17 mol% | 6×10^-7 mol% ocean molar abundance | 0.0014 mol% | 66 mol% | 3×10^-12 mol% crust molar abundance | 0.31 mol% | 3.1 mol% | 5×10^-6 mol% human molar abundance | 12 mol% | 62 mol% |