Input interpretation
sodium hydroxide
Chemical names and formulas

formula | NaOH Hill formula | HNaO name | sodium hydroxide alternate names | aetznatron | ascarite | caustic soda | collo-grillrein | collo-tapetta | lye | soda lye | sodium hydrate | white caustic mass fractions | H (hydrogen) 2.52% | Na (sodium) 57.5% | O (oxygen) 40%
Structure diagram
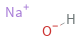
Structure diagram
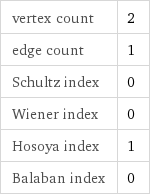
vertex count | 2 edge count | 1 Schultz index | 0 Wiener index | 0 Hosoya index | 1 Balaban index | 0
Basic properties
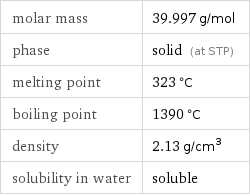
molar mass | 39.997 g/mol phase | solid (at STP) melting point | 323 °C boiling point | 1390 °C density | 2.13 g/cm^3 solubility in water | soluble
Units

Solid properties (at STP)
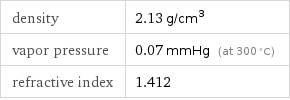
density | 2.13 g/cm^3 vapor pressure | 0.07 mmHg (at 300 °C) refractive index | 1.412
Units

Thermodynamic properties
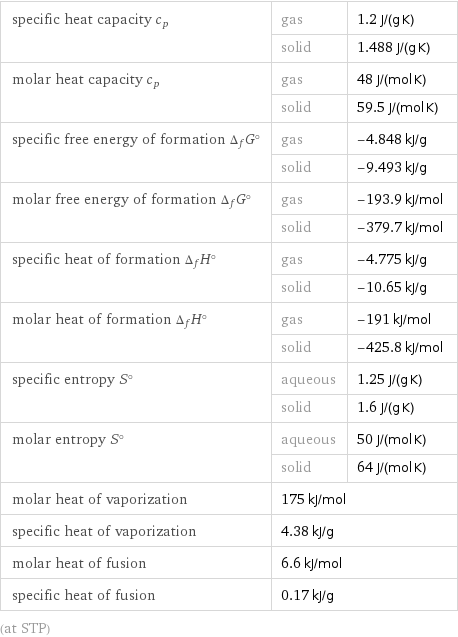
specific heat capacity c_p | gas | 1.2 J/(g K) | solid | 1.488 J/(g K) molar heat capacity c_p | gas | 48 J/(mol K) | solid | 59.5 J/(mol K) specific free energy of formation Δ_fG° | gas | -4.848 kJ/g | solid | -9.493 kJ/g molar free energy of formation Δ_fG° | gas | -193.9 kJ/mol | solid | -379.7 kJ/mol specific heat of formation Δ_fH° | gas | -4.775 kJ/g | solid | -10.65 kJ/g molar heat of formation Δ_fH° | gas | -191 kJ/mol | solid | -425.8 kJ/mol specific entropy S° | aqueous | 1.25 J/(g K) | solid | 1.6 J/(g K) molar entropy S° | aqueous | 50 J/(mol K) | solid | 64 J/(mol K) molar heat of vaporization | 175 kJ/mol | specific heat of vaporization | 4.38 kJ/g | molar heat of fusion | 6.6 kJ/mol | specific heat of fusion | 0.17 kJ/g | (at STP)
Chemical identifiers
![CAS number | 1310-73-2 PubChem CID number | 14798 SMILES identifier | [OH-].[Na+] InChI identifier | InChI=1/Na.H2O/h;1H2/q+1;/p-1/fNa.HO/h;1h/qm;-1 MDL number | MFCD00132264](../image_source/434eda544b2fcb3c67979e6ddab1dacb.png)
CAS number | 1310-73-2 PubChem CID number | 14798 SMILES identifier | [OH-].[Na+] InChI identifier | InChI=1/Na.H2O/h;1H2/q+1;/p-1/fNa.HO/h;1h/qm;-1 MDL number | MFCD00132264
NFPA label

NFPA label

NFPA health rating | 4 NFPA fire rating | 0 NFPA reactivity rating | 4
Toxicity properties

short-term exposure limit | 2 mg/m^3

long-term exposure limit | 2 mg/m^3 (over 8 hours) RTECS classes | agricultural chemical and pesticide | mutagen | primary irritant
Ion equivalents

Na^+ (sodium cation) | 1 (OH)^- (hydroxide anion) | 1