Input interpretation
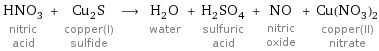
HNO_3 nitric acid + Cu_2S copper(I) sulfide ⟶ H_2O water + H_2SO_4 sulfuric acid + NO nitric oxide + Cu(NO_3)_2 copper(II) nitrate
Chemical names and formulas
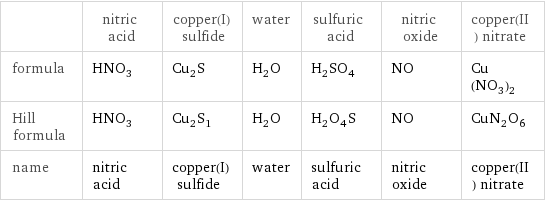
| nitric acid | copper(I) sulfide | water | sulfuric acid | nitric oxide | copper(II) nitrate formula | HNO_3 | Cu_2S | H_2O | H_2SO_4 | NO | Cu(NO_3)_2 Hill formula | HNO_3 | Cu_2S_1 | H_2O | H_2O_4S | NO | CuN_2O_6 name | nitric acid | copper(I) sulfide | water | sulfuric acid | nitric oxide | copper(II) nitrate