Input interpretation
dichlorine hexoxide | copper(II) vanadate
Chemical names and formulas
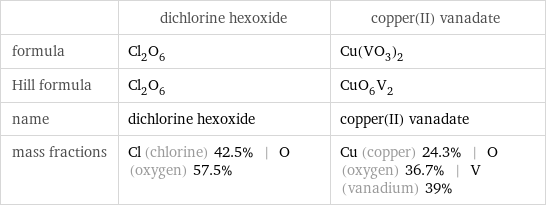
| dichlorine hexoxide | copper(II) vanadate formula | Cl_2O_6 | Cu(VO_3)_2 Hill formula | Cl_2O_6 | CuO_6V_2 name | dichlorine hexoxide | copper(II) vanadate mass fractions | Cl (chlorine) 42.5% | O (oxygen) 57.5% | Cu (copper) 24.3% | O (oxygen) 36.7% | V (vanadium) 39%
Basic properties
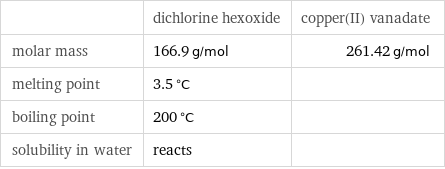
| dichlorine hexoxide | copper(II) vanadate molar mass | 166.9 g/mol | 261.42 g/mol melting point | 3.5 °C | boiling point | 200 °C | solubility in water | reacts |
Units
