Input interpretation

butane | orbital hybridization
Result
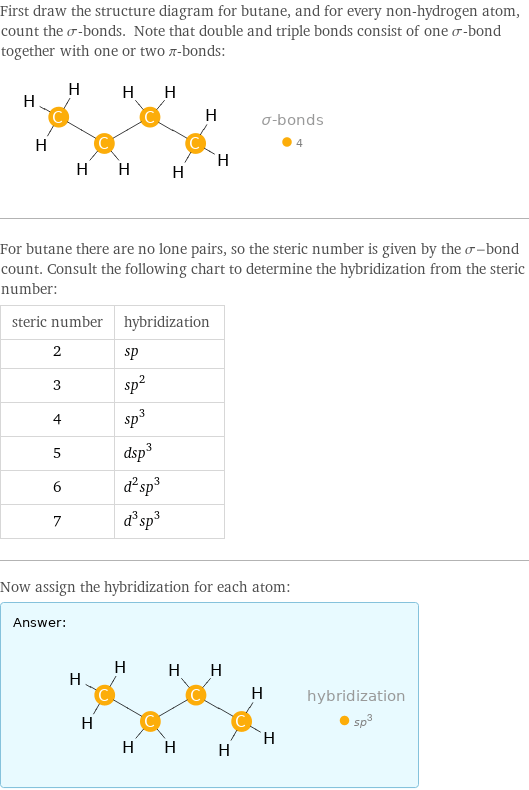
First draw the structure diagram for butane, and for every non-hydrogen atom, count the σ-bonds. Note that double and triple bonds consist of one σ-bond together with one or two π-bonds: For butane there are no lone pairs, so the steric number is given by the σ-bond count. Consult the following chart to determine the hybridization from the steric number: steric number | hybridization 2 | sp 3 | sp^2 4 | sp^3 5 | dsp^3 6 | d^2sp^3 7 | d^3sp^3 Now assign the hybridization for each atom: Answer: | |
Chemical names and formulas

formula | CH_3CH_2CH_2CH_3 Hill formula | C_4H_10 name | butane alternate names | diethyl | Freon 600 | liquefied petroleum gas | n-butane | R-600 mass fractions | C (carbon) 82.7% | H (hydrogen) 17.3%